Difference between revisions of "Basic copper carbonate"
(→Risks) |
|||
| (6 intermediate revisions by 2 users not shown) | |||
| Line 9: | Line 9: | ||
[[[SliderGallery rightalign|basic copper carbonate.jpg~Chemical structure]]] | [[[SliderGallery rightalign|basic copper carbonate.jpg~Chemical structure]]] | ||
| − | == | + | == Risks == |
| − | Insoluble in water and ethanol. Decomposes in acids with the evolution of carbon dioxide bubbles. Turns black with warm alkalis, hydrogen sulfide or sulfur fumes. | + | * Skin contact and inhalation may cause irritation or allergic reactions. |
| + | * Chronic exposure may cause anemia. | ||
| + | * Fisher Scientific: [https://fscimage.fishersci.com/msds/05610.htm MSDS] | ||
| + | |||
| + | == Physical and Chemical Properties == | ||
| + | |||
| + | * Insoluble in water and ethanol. | ||
| + | * Decomposes in acids with the evolution of carbon dioxide bubbles. | ||
| + | * Turns black with warm alkalis, hydrogen sulfide or sulfur fumes. | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 25: | Line 33: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | 200 (dec) | + | | 200 C (dec) |
|- | |- | ||
! scope="row"| Density | ! scope="row"| Density | ||
| − | | 3.7-4.0 | + | | 3.7-4.0 g/ml |
|- | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| mol. wt. = 221.13 | | mol. wt. = 221.13 | ||
|} | |} | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
== Comparisons == | == Comparisons == | ||
| − | [[media: | + | [[media:download_file_436.pdf|Properties of Common Gemstones]] |
| − | |||
| − | |||
| − | |||
| + | [[media:download_file_499.pdf|Characteristics of Common Blue Pigments]] | ||
| − | == | + | == Resources and Citations == |
| + | * R. Gettens, and E. West Fitzhugh, "Azurite and Blue Verditer", ''Artists Pigments'', Vol. 2., A. Roy ed. Oxford University Press, Oxford, 1993. | ||
* R. J. Gettens, G.L. Stout, ''Painting Materials, A Short Encyclopaedia'', Dover Publications, New York, 1966 | * R. J. Gettens, G.L. Stout, ''Painting Materials, A Short Encyclopaedia'', Dover Publications, New York, 1966 | ||
Latest revision as of 10:45, 2 May 2022
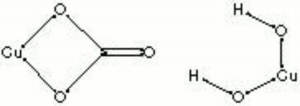
Description
Basic copper carbonate occurs naturally in two crystalline forms, blue (Azurite) and green (Malachite). Azurite and malachite have been used as gemstones and paint pigments since antiquity. They are prepared as pigments by careful selection, grinding, washing, and levigation. As coarsely ground particles, they give a deep opaque color, but when finely ground, the pigments give a lighter more transparent tone. Azurite and malachite are lightfast but are sensitive to acids and sulfur fumes. Basic copper carbonate can also be made artificially by coloring chalk with copper sulfate. The equivalent synthetic pigments, called Blue verditer and Green verditer, tend to have regularly sized particles with rounded edges.
Synonyms and Related Terms
basic cupric carbonate; azurite; malachite; carbonato básico de cobre (Esp.); carbonato basico di rame (It.); carbonato básico de cobre (Port.)
Risks
- Skin contact and inhalation may cause irritation or allergic reactions.
- Chronic exposure may cause anemia.
- Fisher Scientific: MSDS
Physical and Chemical Properties
- Insoluble in water and ethanol.
- Decomposes in acids with the evolution of carbon dioxide bubbles.
- Turns black with warm alkalis, hydrogen sulfide or sulfur fumes.
| Composition | 2CuCO3òCu(OH)2 |
|---|---|
| CAS | 12069-69-1 |
| Mohs Hardness | 3.5 - 4.0 |
| Melting Point | 200 C (dec) |
| Density | 3.7-4.0 g/ml |
| Molecular Weight | mol. wt. = 221.13 |
Comparisons
Properties of Common Gemstones
Characteristics of Common Blue Pigments
Resources and Citations
- R. Gettens, and E. West Fitzhugh, "Azurite and Blue Verditer", Artists Pigments, Vol. 2., A. Roy ed. Oxford University Press, Oxford, 1993.
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 231
- Ralph Mayer, A Dictionary of Art Terms and Techniques, Harper and Row Publishers, New York, 1969 (also 1945 printing)
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- R.D. Harley, Artists' Pigments c. 1600-1835, Butterworth Scientific, London, 1982
- Jack Odgen, Jewellery of the Ancient World, Rizzoli International Publications Inc., New York City, 1982
- Book and Paper Group, Paper Conservation Catalog, AIC, 1984, 1989
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 2697