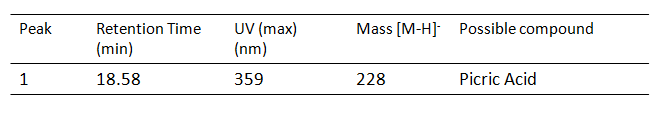Difference between revisions of "CI 10305, Picric Acid, LC"
(Created page with "== Usage History == [http://cameo.mfa.org/wiki/Metanil_yellow Metanil Yellow] == Synonyms == C.I. 13065; C.I. Acid Yellow 36; C.I. Acid Yellow 36 monosodium salt; C.I. Acid...") |
|||
| (16 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
== Usage History == | == Usage History == | ||
| − | [ | + | [https://en.wikipedia.org/wiki/Picric_acid Picric acid] |
== Synonyms == | == Synonyms == | ||
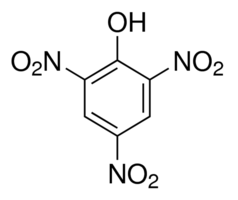
| − | C.I. | + | C.I. 10305; 2,4,6-trinitrophenol; 1,3,5-Trinitrophenol. |
| + | == Molecular Information == | ||
| − | + | [[File:Picric acid safety.PNG|thumb|Picric acid Hazards]] | |
| − | Molecular Formula | + | Molecular Formula C6H3N3O7 |
| − | Molecular Weight | + | Molecular Weight 229.1 g/mol |
| − | [[File: | + | [[File:Picric acid.png|center|frame]] |
== Analytical instrumentation and procedures == | == Analytical instrumentation and procedures == | ||
| − | + | '''Extraction''': The extraction was carried out as follows: a small sample of thread was extracted with the TFA 2M in 1.5ml eppendorfs for 30 min, in 60ºC water bath, with constant agitation. After extraction, each extract was dried in a vacuum system, where the resulting dry residues were reconstituted with 50 μl H2O: MeOH (80:20,v/v). | |
| + | |||
| + | |||
| + | '''HPLC system''': The dye analyses were performed in a Thermofinnigan Surveyor HPLC-DAD system with a Thermofinnigan Surveyor PDA 5 diode array detector (Thermofinnigan, USA), an autosampler and a pump. | ||
| + | |||
| + | '''HPLC Column''': The separations were performed in Zorbax Eclipse Plus C18 (Agilent, USA) with 5μm particle size column (2.1 mm x 150 mm). The column was kept at controlled temperature (35ºC). The samples were injected onto the column via a Rheodyne injector with a 25μl loop. | ||
| − | + | '''LC program''': A solvent gradient of (A) TEA pH= 6,4 3mM and (B) pure methanolwas used at a flow rate of 0.3 mL/min; 0-0,01min A:B (95:5) isocratic, 5 min A:B (90:10) isocratic, 10 min A:B (70:30) isocratic, 15 min A:B (50:50) isocratic, 30 min A:B (45:55) isocratic, 35 min A:B (30:70) isocratic, 45-55 min A:B (5:95) linear, 60-70 min A:B (95:5) linear. | |
| − | + | '''LC-DAD-MSn''': The analyses of dyes were performed on a LC-MS with ProStar 410 autosampler, two 212-LC chromatography pumps, a ProStar 335 diode array detector and a 500-MS ion trap mass spectrometer with an electrospray ionization (ESI) ion source (Varian, Palo Alto, CA, USA). Data acquisition and processing were performed using Varian MS Control 6.9 software.The separations were carried out using a Zorbax Eclipse Plus (Agilent, USA) with 5 μm particle size column (2.1 mm x 150 mm). The column was kept at controlled temperature (35ºC). The samples were injected onto the column via a Rheodyne injector with a 20 μL loop. The gradient adapted from and described in 2.3.1 was used at a flow rate of 0.2 mL/min. The mass spectra were acquired in negative and positive mode depending on the structures. The optimized parameters were as follows: ion spray voltage, 4,8 kV; capillary voltage, 20 V; RF loading 80%. Nitrogen was used as nebulising and drying gas, at a pressure of 35 and 15 psi; drying gas temperature, 300ºC. The multistage MS (MSn) spectra were obtained with an isolation window of 2.0 Da, excitation energy values of 0,9 to 1,5 V and an excitation time of 10 ms. | |
| − | + | [1] | |
= Chromatograms = | = Chromatograms = | ||
| − | [[File: | + | [[File:Picric Acid LC.PNG|center|frame|Absorbance at 347nm (mAU), By Cátia Souto, University of Lisbon, Portugal 2010.[1]]] |
== Results == | == Results == | ||
| − | [[File: | + | [[File:Picric acid result1.PNG|center|frame|compounds identified]] |
== References == | == References == | ||
| − | [1] | + | [1] Cátia Susana da Costa Nogueira Souto, "Analysis of Early Synthetic Dyes with HPLC-DAD-MS An important database for analysis of colorants used in cultural heritage", Thesis, Master Degree in Conservation and Restoration of Textiles. UNIVERSIDADE NOVA DE LISBOA, 2010. |
[[Category:Dye Analysis]] | [[Category:Dye Analysis]] | ||
[[Category:Reference Materials]] | [[Category:Reference Materials]] | ||
[[Category:Synthetic Dyes]] | [[Category:Synthetic Dyes]] | ||
Latest revision as of 09:59, 4 October 2017
Usage History
Synonyms
C.I. 10305; 2,4,6-trinitrophenol; 1,3,5-Trinitrophenol.
Molecular Information
Molecular Formula C6H3N3O7
Molecular Weight 229.1 g/mol
Analytical instrumentation and procedures
Extraction: The extraction was carried out as follows: a small sample of thread was extracted with the TFA 2M in 1.5ml eppendorfs for 30 min, in 60ºC water bath, with constant agitation. After extraction, each extract was dried in a vacuum system, where the resulting dry residues were reconstituted with 50 μl H2O: MeOH (80:20,v/v).
HPLC system: The dye analyses were performed in a Thermofinnigan Surveyor HPLC-DAD system with a Thermofinnigan Surveyor PDA 5 diode array detector (Thermofinnigan, USA), an autosampler and a pump.
HPLC Column: The separations were performed in Zorbax Eclipse Plus C18 (Agilent, USA) with 5μm particle size column (2.1 mm x 150 mm). The column was kept at controlled temperature (35ºC). The samples were injected onto the column via a Rheodyne injector with a 25μl loop.
LC program: A solvent gradient of (A) TEA pH= 6,4 3mM and (B) pure methanolwas used at a flow rate of 0.3 mL/min; 0-0,01min A:B (95:5) isocratic, 5 min A:B (90:10) isocratic, 10 min A:B (70:30) isocratic, 15 min A:B (50:50) isocratic, 30 min A:B (45:55) isocratic, 35 min A:B (30:70) isocratic, 45-55 min A:B (5:95) linear, 60-70 min A:B (95:5) linear.
LC-DAD-MSn: The analyses of dyes were performed on a LC-MS with ProStar 410 autosampler, two 212-LC chromatography pumps, a ProStar 335 diode array detector and a 500-MS ion trap mass spectrometer with an electrospray ionization (ESI) ion source (Varian, Palo Alto, CA, USA). Data acquisition and processing were performed using Varian MS Control 6.9 software.The separations were carried out using a Zorbax Eclipse Plus (Agilent, USA) with 5 μm particle size column (2.1 mm x 150 mm). The column was kept at controlled temperature (35ºC). The samples were injected onto the column via a Rheodyne injector with a 20 μL loop. The gradient adapted from and described in 2.3.1 was used at a flow rate of 0.2 mL/min. The mass spectra were acquired in negative and positive mode depending on the structures. The optimized parameters were as follows: ion spray voltage, 4,8 kV; capillary voltage, 20 V; RF loading 80%. Nitrogen was used as nebulising and drying gas, at a pressure of 35 and 15 psi; drying gas temperature, 300ºC. The multistage MS (MSn) spectra were obtained with an isolation window of 2.0 Da, excitation energy values of 0,9 to 1,5 V and an excitation time of 10 ms.
[1]
Chromatograms
Results
References
[1] Cátia Susana da Costa Nogueira Souto, "Analysis of Early Synthetic Dyes with HPLC-DAD-MS An important database for analysis of colorants used in cultural heritage", Thesis, Master Degree in Conservation and Restoration of Textiles. UNIVERSIDADE NOVA DE LISBOA, 2010.