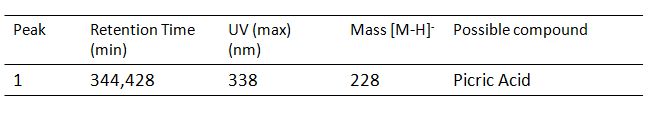CI 10305, Picric Acid, LC
Usage History
Synonyms
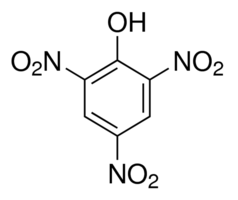
C.I. 10305; 2,4,6-trinitrophenol; 1,3,5-Trinitrophenol.
Molecular Information
Molecular Formula C6H3N3O7
Molecular Weight 229.1 g/mol
Analytical instrumentation and procedures
Dyes were extracted from textiles (silk or wool) using formic acid/methanol (5:95, v/v).
HPLC column: Vydac 214TP52 analytical column (2.1 mm diameterX250 mm; 5-ím particle size).
HPLC-DAD-MS instrument: HPLC-DAD-MS analysis was performed with an Agilent 1100 liquid chromatography system consisting of an automatic injector, a gradient pump, a HP series 1100 DAD, and an Agilent series 1100 VL on-line atmospheric pressure ionization electrospray ionization mass spectrometer.
HPLC gradient profile: Separations were done on a The column was eluted at a flow rate of 0.2 mL/min with a tertiary gradient of water (A),acetonitrile (B), and 1% (v/v) aqueous formic acid (C) with the following elution program: 0 min, 90% A, 5% B, 5% C; 0-55 min, a linear gradient to 35% A, 60% B, 5% C; 55-60 min, a linear gradient elution to 15% A, 80% B, 5% C; 60-62 min, isocratic elution at 15% A, 80% B, 5% C; 62-70 min gradient elution to 90% A, 5% B, 5% C; and reequilibration with the latter solvent for 15 min. The mass spectrometer was run both in the negative ion mode because metanil Yellow is negatively charged without Na+.
Chromatograms
Results
References
[1] Cátia Susana da Costa Nogueira Souto, "Analysis of Early Synthetic Dyes with HPLC-DAD-MS An important database for analysis of colorants used in cultural heritage", Thesis, Master Degree in Conservation and Restoration of Textiles. UNIVERSIDADE NOVA DE LISBOA, 2010.