Difference between revisions of "Cobalt violet"
| Line 8: | Line 8: | ||
cobalt arsenate (light); cobalt phosphate (deep); Kobaltviolett (Deut.); violeta de cobalto (Esp.); violet de cobalt (Fr.); violetto di cobalto (It.); violeta de cobalto (Port.) | cobalt arsenate (light); cobalt phosphate (deep); Kobaltviolett (Deut.); violeta de cobalto (Esp.); violet de cobalt (Fr.); violetto di cobalto (It.); violeta de cobalto (Port.) | ||
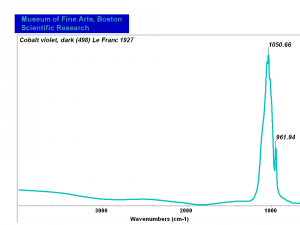
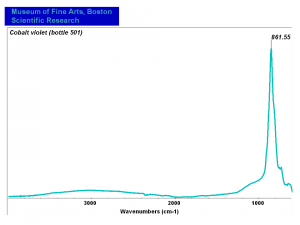
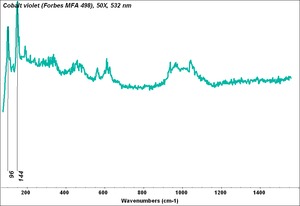
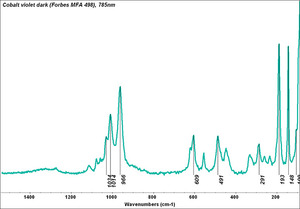
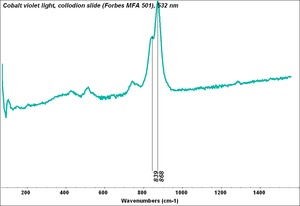
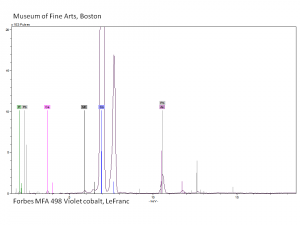
| − | [[[SliderGallery rightalign|Cobalt Violet, dark (498).PNG ~FTIR|Cobalt Violet(501).PNG~FTIR|Cobalt violet (Forbes MFA 498), 50X, 532 nm.TIF~Raman (MFA) (532nm)|Cobalt violet dark (Forbes MFA 498), 785nm resize.tif~Raman (MFA) (785nm)|Slide11_F498.PNG~XRF]]] | + | [[[SliderGallery rightalign|Cobalt Violet, dark (498).PNG ~FTIR (MFA) (Forbes 485)|Cobalt Violet(501).PNG~FTIR (MFA) (Forbes 501)|Cobalt violet (Forbes MFA 498), 50X, 532 nm.TIF~Raman (MFA) (532nm)|Cobalt violet dark (Forbes MFA 498), 785nm resize.tif~Raman (MFA) (785nm)|Cobalt violet light, collodion slide (Forbes MFA 501), 532 nm.TIF~Raman (MFA) (532nm)|Slide11_F498.PNG~XRF]]] |
== Hazards and Safety == | == Hazards and Safety == | ||
Revision as of 11:19, 10 October 2019
Description
A general name for several violet color cobalt pigments. First developed in 1859, cobalt violet was the primary permanent violet pigment available. Cobalt violets range from deep to pale shades with either a pink or blue hue. The first cobalt violets used were composed of cobalt arsenate. This highly toxic compound is now rarely used. Instead most current cobalt violets are nontoxic and are made from either cobalt phosphate, or cobalt ammonium phosphate. Cobalt violets are used in paints, glass, glazes and enamels.
Synonyms and Related Terms
cobalt arsenate (light); cobalt phosphate (deep); Kobaltviolett (Deut.); violeta de cobalto (Esp.); violet de cobalt (Fr.); violetto di cobalto (It.); violeta de cobalto (Port.)
Hazards and Safety
Skin contact may cause allergies, especially on elbows, neck and ankles. Chronic inhalation may cause asthma. Ingestion may cause vomiting, diarrhea and the sensation of hotness.
Additional Information
° Pigments Through the Ages: Cobalt violet
° Corbeil, Marie-Claude, Jean-Pierre Charland, Elizabeth Moffatt. 'The characterization of cobalt violet pigments' Studies in Conservation vol.47 (2002), pp.237-249.
Sources Checked for Data in Record
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966
- Ralph Mayer, A Dictionary of Art Terms and Techniques, Harper and Row Publishers, New York, 1969 (also 1945 printing)
- Encyclopedia Britannica, http://www.britannica.com Comment: "Cobalt Processing." Encyclopædia Britannica. 2004. Encyclopædia Britannica Premium Service. 15 Apr. 2004 .
- Website address 1 Comment: http://webexhibits.org/pigments/indiv/overview/coviolet.html - developed 1859
- Thomas B. Brill, Light Its Interaction with Art and Antiquities, Plenum Press, New York City, 1980
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- Art and Architecture Thesaurus Online, http://www.getty.edu/research/tools/vocabulary/aat/, J. Paul Getty Trust, Los Angeles, 2000