Difference between revisions of "Fructose"
Jump to navigation
Jump to search
m (Text replace - "== Authority ==" to "== Sources Checked for Data in Record ==") |
|||
| (2 intermediate revisions by the same user not shown) | |||
| Line 8: | Line 8: | ||
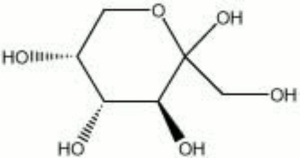
[[[SliderGallery rightalign|fructose.jpg~Chemical structure]]] | [[[SliderGallery rightalign|fructose.jpg~Chemical structure]]] | ||
| + | == Risks == | ||
| − | == | + | * Combustible. |
| + | * ThermoFisher: [https://www.fishersci.com/store/msds?partNumber=AC161350010&productDescription=D-%28-%29-FRUCTOSE%2C+98%25+1KG&vendorId=VN00032119&countryCode=US&language=en SDS] | ||
| + | ==Physical and Chemical Properties == | ||
Soluble in water, ethanol, ether, hot acetone, pyridine, and ethylamine. | Soluble in water, ethanol, ether, hot acetone, pyridine, and ethylamine. | ||
| Line 22: | Line 25: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | 103-105(dec) | + | | 103-105 C (dec) |
|- | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| Line 28: | Line 31: | ||
|} | |} | ||
| − | == | + | ==Resources and Citations == |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
* G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 368 | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 368 | ||
Latest revision as of 13:58, 26 August 2022
Description
A sugar that occurs naturally in fruits and Honey. Fructose forms orthorhombic white crystals when dried from water or alcohol solutions. It is the sweetest of all sugars. Fructose is used as a preservative and for flavoring in food.
Synonyms and Related Terms
fruit sugar; levulose; d-fructopyranose; d-fructose
Risks
- Combustible.
- ThermoFisher: SDS
Physical and Chemical Properties
Soluble in water, ethanol, ether, hot acetone, pyridine, and ethylamine.
| Composition | C6H12O6 |
|---|---|
| CAS | 57-48-7 |
| Melting Point | 103-105 C (dec) |
| Molecular Weight | mol. wt. = 180.16 |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 368
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 4295
- The American Heritage Dictionary or Encarta, via Microsoft Bookshelf 98, Microsoft Corp., 1998