Difference between pages "Kiyomitsu I, Actors Ôtani Hiroji III as Okabe no Rokuyata and Nakamura Sukegorô II as Inomata no Koheiroku, 46.1409" and "Sodium formate"
(Difference between pages)
Jump to navigation
Jump to search
| Line 1: | Line 1: | ||
| − | + | == Description == | |
| − | + | White, slightly [[hygroscopic|hygroscopic]] crystals. Sodium formate is used as a neutral [[buffer|buffering agent]]. It is also used as a [[sequestrant|sequestering agent]] for trivalent metal ions, such as [[chromium|chromium]]. Sodium formate is added to increase the fixation in chrome tanning baths. | |
| − | + | == Synonyms and Related Terms == | |
| − | + | Protan [Hercules] | |
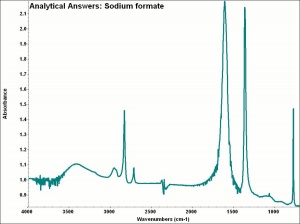
| − | + | [[[SliderGallery rightalign|aaiNAFORMATE.jpg~FTIR]]] | |
| + | ==Risks== | ||
| + | * ThermoFisher: [https://www.fishersci.com/store/msds?partNumber=S648500&productDescription=SODIUM+FORMATE+CERTIFIED+500G&vendorId=VN00033897&countryCode=US&language=en SDS] | ||
| − | + | == Physical and Chemical Properties == | |
| − | + | Soluble in water (pH=7) and glycerol. Slightly soluble in ethanol. Insoluble in ether. | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| − | ! | + | ! scope="row"| Composition |
| + | | CHOONa | ||
|- | |- | ||
| − | | | + | ! scope="row"| CAS |
| + | | 141-53-7 | ||
|- | |- | ||
| − | | | + | ! scope="row"| Melting Point |
| + | | 253 C | ||
|- | |- | ||
| − | | | + | ! scope="row"| Density |
| + | | 1.919 g/ml | ||
|- | |- | ||
| − | | | + | ! scope="row"| Molecular Weight |
| − | + | | mol. wt. = 68.0 | |
| − | | | ||
| − | |||
| − | |||
|} | |} | ||
| − | |||
| − | + | ==Resources and Citations== | |
| − | + | ||
| − | + | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 343 | |
| − | + | ||
| − | + | * Richard S. Lewis, ''Hawley's Condensed Chemical Dictionary'', Van Nostrand Reinhold, New York, 10th ed., 1993 | |
| − | [[Category: | + | |
| + | * Matt Roberts, Don Etherington, ''Bookbinding and the Conservation of Books: a Dictionary of Descriptive Terminology'', U.S. Government Printing Office, Washington DC, 1982 | ||
| + | |||
| + | * ''Van Nostrand's Scientific Encyclopedia'', Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976 | ||
| + | |||
| + | * ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 8765 | ||
| + | |||
| + | |||
| + | |||
| + | [[Category:Materials database]] | ||
Latest revision as of 08:48, 2 June 2022
Description
White, slightly Hygroscopic crystals. Sodium formate is used as a neutral buffering agent. It is also used as a sequestering agent for trivalent metal ions, such as Chromium. Sodium formate is added to increase the fixation in chrome tanning baths.
Synonyms and Related Terms
Protan [Hercules]
Risks
- ThermoFisher: SDS
Physical and Chemical Properties
Soluble in water (pH=7) and glycerol. Slightly soluble in ethanol. Insoluble in ether.
| Composition | CHOONa |
|---|---|
| CAS | 141-53-7 |
| Melting Point | 253 C |
| Density | 1.919 g/ml |
| Molecular Weight | mol. wt. = 68.0 |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 343
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Matt Roberts, Don Etherington, Bookbinding and the Conservation of Books: a Dictionary of Descriptive Terminology, U.S. Government Printing Office, Washington DC, 1982
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 8765