Difference between revisions of "Sodium carbonate"
Jump to navigation
Jump to search
| Line 2: | Line 2: | ||
White, odorless crystals. Sodium carbonate has three distinct hydrated forms. The monohydrate is commonly called sodium carbonate while anhydrous sodium carbonate is called [[soda%20ash|soda ash]], and sodium carbonate decahydrate is called [[sal%20soda|sal soda]], or natron. Anhydrous sodium carbonate occurs in nature as the mineral thermonatrite. It forms a strongly alkaline aqueous solution and is used in the manufacture of [[leather|leather]] and [[paper|paper]]. Sodium carbonate is used as an ingredient in some types of [[glass|glass]], [[detergent|detergents]], [[bleaching%20agent|bleaches]], and [[water%20softener|water softeners]]. It is also used in photographic [[developer|developing]] baths and textiles [[dye|dye]] baths. | White, odorless crystals. Sodium carbonate has three distinct hydrated forms. The monohydrate is commonly called sodium carbonate while anhydrous sodium carbonate is called [[soda%20ash|soda ash]], and sodium carbonate decahydrate is called [[sal%20soda|sal soda]], or natron. Anhydrous sodium carbonate occurs in nature as the mineral thermonatrite. It forms a strongly alkaline aqueous solution and is used in the manufacture of [[leather|leather]] and [[paper|paper]]. Sodium carbonate is used as an ingredient in some types of [[glass|glass]], [[detergent|detergents]], [[bleaching%20agent|bleaches]], and [[water%20softener|water softeners]]. It is also used in photographic [[developer|developing]] baths and textiles [[dye|dye]] baths. | ||
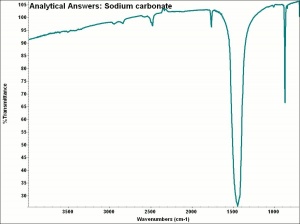
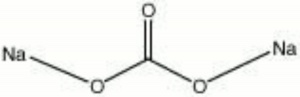
| − | + | [[[SliderGallery rightalign|aaiNACO3.jpg~FTIR|sodium carbonate.jpg~Chemical structure]]] | |
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
soda; sodium carbonate monohydrate; soda monohydrate; soda crystals; calcined soda ash | soda; sodium carbonate monohydrate; soda monohydrate; soda crystals; calcined soda ash | ||
| − | |||
== Risks == | == Risks == | ||
| − | Noncombustible. Toxic by ingestion and inhalation. | + | * Noncombustible. |
| − | + | * Toxic by ingestion and inhalation. | |
| − | Corrosive. Skin contact will cause burns. | + | * Corrosive. |
| + | * Skin contact will cause burns. | ||
| + | * ThermoFisher: [https://beta-static.fishersci.com/content/dam/fishersci/en_US/documents/programs/education/regulatory-documents/sds/chemicals/chemicals-s/S25539D.pdf SDS] | ||
| − | |||
== Physical and Chemical Properties == | == Physical and Chemical Properties == | ||
| Line 28: | Line 28: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | 109 | + | | 109 C |
|- | |- | ||
! scope="row"| Density | ! scope="row"| Density | ||
| − | | 1.55 | + | | 1.55 g/ml |
|- | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| Line 64: | Line 64: | ||
* ''The American Heritage Dictionary'' or ''Encarta'', via Microsoft Bookshelf 98, Microsoft Corp., 1998 | * ''The American Heritage Dictionary'' or ''Encarta'', via Microsoft Bookshelf 98, Microsoft Corp., 1998 | ||
| − | * | + | * Photographic chemicals: www.jetcity.com/~mrjones/chemdesc.htm |
* ''CRC Handbook of Chemistry and Physics'', Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index = 1.415, 1.535, 1.546 | * ''CRC Handbook of Chemistry and Physics'', Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index = 1.415, 1.535, 1.546 | ||
Latest revision as of 14:59, 1 June 2022
Description
White, odorless crystals. Sodium carbonate has three distinct hydrated forms. The monohydrate is commonly called sodium carbonate while anhydrous sodium carbonate is called Soda ash, and sodium carbonate decahydrate is called Sal soda, or natron. Anhydrous sodium carbonate occurs in nature as the mineral thermonatrite. It forms a strongly alkaline aqueous solution and is used in the manufacture of Leather and Paper. Sodium carbonate is used as an ingredient in some types of Glass, detergents, bleaches, and water softeners. It is also used in photographic developing baths and textiles Dye baths.
Synonyms and Related Terms
soda; sodium carbonate monohydrate; soda monohydrate; soda crystals; calcined soda ash
Risks
- Noncombustible.
- Toxic by ingestion and inhalation.
- Corrosive.
- Skin contact will cause burns.
- ThermoFisher: SDS
Physical and Chemical Properties
Soluble in water, glycerol. Insoluble in ethanol.
| Composition | NaCO3 - H2O |
|---|---|
| CAS | 497-19-8 |
| Melting Point | 109 C |
| Density | 1.55 g/ml |
| Molecular Weight | mol. wt. = 105.99 |
| Refractive Index | 1.415, 1.535, 1.546 |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 734
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Hermann Kuhn, Conservation and Restoration of Works of Art and Antiquities, Butterworths, London, 1986
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- Matt Roberts, Don Etherington, Bookbinding and the Conservation of Books: a Dictionary of Descriptive Terminology, U.S. Government Printing Office, Washington DC, 1982
- John and Margaret Cannon, Dye Plants and Dyeing, Herbert Press, London, 1994
- The Dictionary of Paper, American Paper Institute, New York, Fourth Edition, 1980
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 8739
- The American Heritage Dictionary or Encarta, via Microsoft Bookshelf 98, Microsoft Corp., 1998
- Photographic chemicals: www.jetcity.com/~mrjones/chemdesc.htm
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index = 1.415, 1.535, 1.546