Difference between pages "File:Africano marble MFA49476.jpg" and "Barium nitrate"
(Difference between pages)
Jump to navigation
Jump to search
(→Risks) |
|||
| Line 1: | Line 1: | ||
| − | + | == Description == | |
| + | [[[SliderGallery rightalign|barium nitrate.jpg~Chemical structure]]] | ||
| + | White crystals or fused mass that is a strong oxidizing agent. . Barium nitrate is primarily used to produce green fireworks and green signal lights. It is also used in the manufacture of glass and ceramic glazes as well as for a rodenticide. | ||
| + | |||
| + | == Risks == | ||
| + | |||
| + | * Highly toxic by ingestion. | ||
| + | * Contact with skin and membranes may cause irritation. | ||
| + | * ThermoFisher: [https://www.fishersci.com/store/msds?partNumber=AC453800100&productDescription=BARIUM+NITRATE%2C+PURE+10KG&vendorId=VN00032119&countryCode=US&language=en SDS] | ||
| + | |||
| + | ==Physical and Chemical Properties== | ||
| + | |||
| + | Soluble in water. Insoluble in ethanol. | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | ! scope="row"| Composition | ||
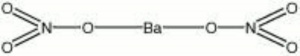
| + | | Ba(NO3)2 | ||
| + | |- | ||
| + | ! scope="row"| CAS | ||
| + | | 10022-31-8 | ||
| + | |- | ||
| + | ! scope="row"| Melting Point | ||
| + | | 575 C | ||
| + | |- | ||
| + | ! scope="row"| Density | ||
| + | | 3.244 g/ml | ||
| + | |- | ||
| + | ! scope="row"| Molecular Weight | ||
| + | | mol. wt. = 261.34 | ||
| + | |} | ||
| + | |||
| + | ==Resources and Citations== | ||
| + | |||
| + | * Richard S. Lewis, ''Hawley's Condensed Chemical Dictionary'', Van Nostrand Reinhold, New York, 10th ed., 1993 Comment: melting point=575C | ||
| + | |||
| + | * ''Van Nostrand's Scientific Encyclopedia'', Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976 | ||
| + | |||
| + | * ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 1012 | ||
| + | |||
| + | * Wikipedia: http://en.wikipedia.org/wiki/Barium_nitrate (Accessed Jan. 6, 2006) has melting point = 595C | ||
| + | |||
| + | |||
| + | |||
| + | [[Category:Materials database]] | ||
Latest revision as of 09:42, 2 May 2022
Description
White crystals or fused mass that is a strong oxidizing agent. . Barium nitrate is primarily used to produce green fireworks and green signal lights. It is also used in the manufacture of glass and ceramic glazes as well as for a rodenticide.
Risks
- Highly toxic by ingestion.
- Contact with skin and membranes may cause irritation.
- ThermoFisher: SDS
Physical and Chemical Properties
Soluble in water. Insoluble in ethanol.
| Composition | Ba(NO3)2 |
|---|---|
| CAS | 10022-31-8 |
| Melting Point | 575 C |
| Density | 3.244 g/ml |
| Molecular Weight | mol. wt. = 261.34 |
Resources and Citations
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993 Comment: melting point=575C
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 1012
- Wikipedia: http://en.wikipedia.org/wiki/Barium_nitrate (Accessed Jan. 6, 2006) has melting point = 595C
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 17:28, 1 May 2022 |  | 1,211 × 1,600 (139 KB) | MDerrick (talk | contribs) |
You cannot overwrite this file.
File usage
The following page uses this file: