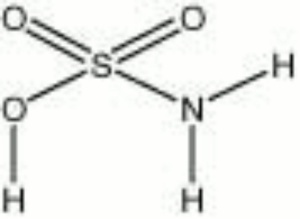
Sulfamic acid
Jump to navigation
Jump to search
Description
White, crystalline solid. Sulfamic acid is used as a flame retardant for textiles and wood, buffer, acid cleaner, chlorine stabilizer in swimming pools, nitrite scavenger, and sulfonating agent. It is also used as a Bleaching agent for paper pulp, textiles, and color photographs.
Synonyms and Related Terms
amidosulfonic acid; amidosulfuric acid; sulfamidic acid
Risks
- Toxic by ingestion.
- Corrosive to eyes, skin and lungs causing irritation and burns.
- Heating results in the production of toxic sulfur dioxide fumes.
- ThermoFisher: SDS
Physical and Chemical Properties
Soluble in water, but hydrolyzed in water to form ammonium bisulfate. Slightly soluble in organic solvents.
| Composition | SOOHNH2 |
|---|---|
| CAS | 5329-14-6 |
| Melting Point | 205 C (dec) |
| Density | 2.15 g/ml |
| Molecular Weight | mol. wt. = 97.1 |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 782
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- The Dictionary of Paper, American Paper Institute, New York, Fourth Edition, 1980
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 9090