Difference between revisions of "Brucite"
Jump to navigation
Jump to search
| Line 15: | Line 15: | ||
* EChemi.com: [https://www.echemi.com/sds/brucite-mgoh2-pd180727138644.html SDS] | * EChemi.com: [https://www.echemi.com/sds/brucite-mgoh2-pd180727138644.html SDS] | ||
| − | == | + | ==Physical and Chemical Properties== |
| − | Tabular, rhombohedron crystals, may sometimes be fibrous. Perfect cleavage parallel to prism base. Luster = waxy to vitreous. Streak = white. | + | * Tabular, rhombohedron crystals, may sometimes be fibrous. |
| + | * Perfect cleavage parallel to prism base. | ||
| + | * Luster = waxy to vitreous. | ||
| + | * Streak = white. | ||
{| class="wikitable" | {| class="wikitable" | ||
Revision as of 11:32, 10 May 2022
Description
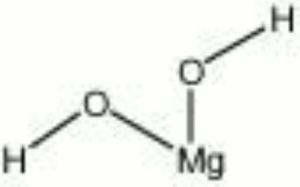
A white to gray mineral composed of Magnesium hydroxide. Brucite was named for Archibald Bruce, an American mineralogist in the late 18th and early 19th centuries. It occurs naturally in deposits, often with Serpentine and Dolomite, in Italy (Teulada), Sweden (Jakobsberg, Filipstad, Nordmark), Canada, and the U.S. (Nevada, New Jersey, Pennsylvania, Texas). The soft mineral can be transparent to translucent with a pearly luster. Brucite is used as a refractory material as well as for a source of Magnesium metal and Magnesia.
Synonyms and Related Terms
nemalite; magnesium hydroxide; magnesium hydrate; milk of magnesia; brucita (Esp.); brucite (Port.); Brucit (Deut.); bruciet (Ned.)
Risks
- EChemi.com: SDS
Physical and Chemical Properties
- Tabular, rhombohedron crystals, may sometimes be fibrous.
- Perfect cleavage parallel to prism base.
- Luster = waxy to vitreous.
- Streak = white.
| Composition | Mg(OH)2 |
|---|---|
| CAS | 1309-42-8 |
| Mohs Hardness | 2.5 |
| Density | 2.39 g/ml |
| Molecular Weight | mol. wt. = 58.32 |
Resources and Citations
- Mineralogy Database: Brucite
- Encyclopedia Britannica, http://www.britannica.com Comment: "brucite" [Accessed December 4, 2001].
- C.W.Chesterman, K.E.Lowe, Audubon Society Field Guide to North American Rocks and Minerals, Alfred A. Knopf, New York, 1979
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Wikipedia: http://en.wikipedia.org/wiki/Brucite (Accessed Sept. 2, 2005)
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- MSDS Sheet Comment: density - 2.36