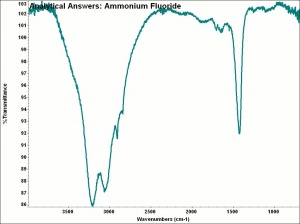
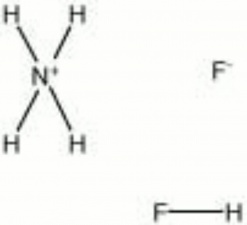
Ammonium bifluoride
Jump to navigation
Jump to search
Description
White, deliquescent crystals that are corrosive to most materials. Ammonium bifluoride is used as an etchant for Glass and Aluminum. It is also used to sterilizing brewing and dairy equipment.
Synonyms and Related Terms
ammonium acid fluoride; ammonium hydrogen fluoride; white acid
Risks
- Corrosive to skin, eyes and lungs. Toxic by ingestion.
- IntegraChem: SDS
Physical and Chemical Properties
Soluble in water, ethanol. pH=3.5
| Composition | NH4HF2 |
|---|---|
| CAS | 1341-49-7 |
| Melting Point | 124.6 C |
| Density | 1.211 g/ml |
| Molecular Weight | mol. wt. = 57.04 |
| Boiling Point | 240 C |
Resources and Citations
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 299
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 523