Difference between revisions of "Ferric sulfate"
Jump to navigation
Jump to search
(→Risks) |
|||
| Line 13: | Line 13: | ||
* Non-combustible. | * Non-combustible. | ||
* Decomposes with light | * Decomposes with light | ||
| + | * Respiratory system irritant | ||
* Fisher Scientific: [https://beta-static.fishersci.com/content/dam/fishersci/en_US/documents/programs/education/regulatory-documents/sds/chemicals/chemicals-f/S25322A.pdf SDS] | * Fisher Scientific: [https://beta-static.fishersci.com/content/dam/fishersci/en_US/documents/programs/education/regulatory-documents/sds/chemicals/chemicals-f/S25322A.pdf SDS] | ||
| + | |||
==Physical and Chemical Properties=== | ==Physical and Chemical Properties=== | ||
Latest revision as of 11:51, 9 December 2022
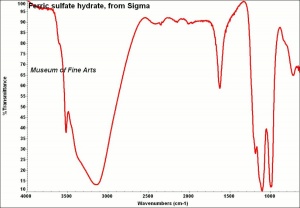
Description
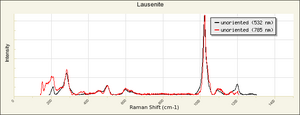
Grayish-white powder formed by adding Sulfuric acid to Ferric hydroxide. Ferric sulfate is very Hygroscopic. It is used as a mordant in textile dyeing and as a component in iron gall inks. Ferric sulfate is also used in water purification systems. Ferric sulfate occurs naturally in minerals with varying states of hydration, including lausenite [Fe2(SO4)3-5H2O], Kornelite [Fe2(SO4)3-7H2O], Coquimbite [Fe2(SO4)3-9H2O], and Quenstedtite [Fe2(SO4)3-10H2O].
Synonyms and Related Terms
iron III sulfate; ferric sulphate (Br.); ferric persulfate; ferric sesquisulfate; ferric tersulfate
Risks
- Non-combustible.
- Decomposes with light
- Respiratory system irritant
- Fisher Scientific: SDS
Physical and Chemical Properties=
Slightly soluble in water and alcohol. Insoluble in organic solvents.
| Composition | Fe2(SO4)3 |
|---|---|
| CAS | 10028-22-5 |
| Melting Point | 480 C (d) |
| Density | 2.0-2.1 g/ml |
| Molecular Weight | 399.88 |
Resources and Citations
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Merck Index, Susan Budavari (ed.), Merck Research Labs, Whitehouse Station, NJ, 12th Edition, 1996 Comment: entry 3963