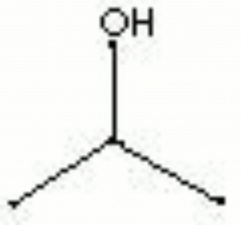
Isopropyl alcohol
Revision as of 13:09, 27 April 2013 by (username removed)
Description
Colorless liquid with a pleasant smell. Isopropyl alcohol, or isopropanol, is used as a solvent for gums, shellac, oil nondrying oils, resin natural resins, and inks. It is also used as an antiseptic in cleansers and body lotions. Isopropanol is added to alcohol ethanol as a alcohol denaturant.
Synonyms and Related Terms
isopropanol; rubbing alcohol; IPA; dimethylcarbinol; sec-propyl alcohol; 2-propanol
Other Properties
Miscible with water, ethanol, ether, chloroform. Insoluble in salt solutions.
| Composition | (CH3)2CHOH |
|---|---|
| CAS | 67-63-0 |
| Melting Point | -86 |
| Density | 0.7863 |
| Molecular Weight | mol. wt.=60.1 |
| Refractive Index | 1.3756 |
| Boiling Point | 82.4 |
Hazards and Safety
Flammable. Dangerous fire risk. Flash point = 12 C (54 F)
Skin contact may cause dryness and irritation. Toxic by ingestion and inhalation.
Mallinckrodt Baker: MSDS
Comparisons
Authority
- Richard S. Lewis, Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5227; ref. index=1.3756
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index=1.375