Difference between revisions of "Methyl methacrylate"
(username removed) |
(username removed) |
||
| Line 1: | Line 1: | ||
== Description == | == Description == | ||
| − | A colorless, volatile liquid that is the monomer for [http://cameo.mfa.org/materials/fullrecord.asp?name=polymethyl | + | A colorless, volatile liquid that is the monomer for [http://cameo.mfa.org/materials/fullrecord.asp?name=polymethyl%20methacrylate polymethyl methacrylate] resins. Methyl methacrylate forms a clear glasslike, thermoplastic polymer that is light weight but hard to break. Methyl methacrylate polymerizes with heat, light, ionizing radiation, or catalysts. It can be copolymerized with many other monomers including other acrylate and methacrylate esters. |
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| − | methacrylic acid methyl ester; methyl 2-methylpropenoate; metacrilato de metilo (Esp.); 2-metilpropenoato de metilo (Esp.); | + | methacrylic acid methyl ester; methyl 2-methylpropenoate; metacrilato de metilo (Esp.); 2-metilpropenoato de metilo (Esp.); méthylméthacrylate (Fr.); metilmetacrilato (It.); acrilato de metilo (Port.) |
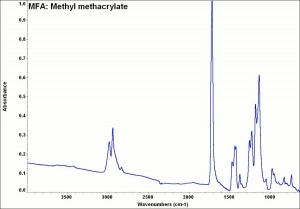
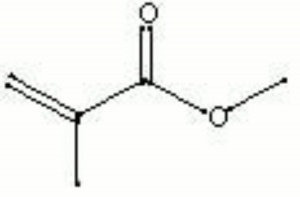
[[[SliderGallery rightalign|MFA- Methyl methacrylate.jpg~FTIR|methyl methacrylate.jpg~Chemical structure]]] | [[[SliderGallery rightalign|MFA- Methyl methacrylate.jpg~FTIR|methyl methacrylate.jpg~Chemical structure]]] | ||
| Line 47: | Line 47: | ||
== Authority == | == Authority == | ||
| − | * | + | * R. J. Gettens, G.L. Stout, ''Painting Materials, A Short Encyclopaedia'', Dover Publications, New York, 1966 |
| − | * | + | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 10 |
| − | * | + | * Richard S. Lewis, ''Hawley's Condensed Chemical Dictionary'', Van Nostrand Reinhold, New York, 10th ed., 1993 |
| − | * | + | * Theodore J. Reinhart, 'Glossary of Terms', ''Engineered Plastics'', ASM International, 1988 |
| − | * | + | * Random House, ''Webster's Encyclopedic Unabridged Dictionary of the English Language'', Grammercy Book, New York, 1997 |
[[Category:Materials database]] | [[Category:Materials database]] | ||
Revision as of 07:53, 24 July 2013
Description
A colorless, volatile liquid that is the monomer for polymethyl methacrylate resins. Methyl methacrylate forms a clear glasslike, thermoplastic polymer that is light weight but hard to break. Methyl methacrylate polymerizes with heat, light, ionizing radiation, or catalysts. It can be copolymerized with many other monomers including other acrylate and methacrylate esters.
Synonyms and Related Terms
methacrylic acid methyl ester; methyl 2-methylpropenoate; metacrilato de metilo (Esp.); 2-metilpropenoato de metilo (Esp.); méthylméthacrylate (Fr.); metilmetacrilato (It.); acrilato de metilo (Port.)
Other Properties
Soluble in most organic solvents. Slightly soluble in water.
| Composition | CH2:C(CH3)COOCH3 |
|---|---|
| CAS | 80-62-6 |
| Melting Point | -48 |
| Density | 0.940 |
| Molecular Weight | mol. wt. = 100.1 |
| Refractive Index | 1.482-1.521 |
| Boiling Point | 100-101 |
Hazards and Safety
Highly flammable. Flash point = 10 C. Explosive limits in air 2.1-12.5%.
Hazardous by ingestion, inhalation and skin absorption. Contact causes irritation.
LINK: International Chemical Safety Card
Authority
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 10
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Theodore J. Reinhart, 'Glossary of Terms', Engineered Plastics, ASM International, 1988
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997