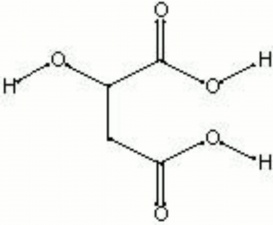
Malic acid
Jump to navigation
Jump to search
Description
White, water-soluble crystals that occur naturally in apples and other fruits. Malic acid is used as a chelating and buffering agent. The weak acid is also used to acidify food and to age wine.
Synonyms and Related Terms
apple acid; hydroxysuccinic acid; hydroxybutanedioic acid
Risks
- Combustible.
- Contact may cause irritation.
- ThermoFisher: SDS
Physical and Chemical Properties
Soluble in water, methanol, diethyl ether, acetone and ethanol. Slightly soluble in ether. Insoluble in benzene.
pH = 2.2 (0.1 N solution)
| Composition | COOHCH2CH(OH)COOH |
|---|---|
| CAS | 617-48-1 (dl form) |
| Melting Point | 131-132 C |
| Density | 1.601 g/ml |
| Molecular Weight | mol. wt. = 134.09 |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 55
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5747
- Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, Technology and Conservation, Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: pH = 2.2 (0.1 N solution)