Difference between revisions of "Trichloroethylene"
Jump to navigation
Jump to search
m (Text replace - "\[http:\/\/cameo\.mfa\.org\/materials\/fullrecord\.asp\?name=([^\s]+)\s(.*)\]" to "$2") |
|||
| (One intermediate revision by the same user not shown) | |||
| Line 1: | Line 1: | ||
== Description == | == Description == | ||
| − | Colorless liquid with a chloroform odor. Trichloroethylene has been used as a solvent for [[fat|fats]], [[oil|oils]], [[wax|waxes]], [[natural%20resin|resins]], [[rubber | + | Colorless liquid with a chloroform odor. Trichloroethylene has been used as a solvent for [[fat|fats]], [[oil|oils]], [[wax|waxes]], [[natural%20resin|resins]], [[rubber|rubber]], [[plastic|plastics]], [[paint|paints]], and [[varnish|varnishes]]. It was widely used for degreasing and dry-cleaning, but by 1990 its usage was banned in many states. |
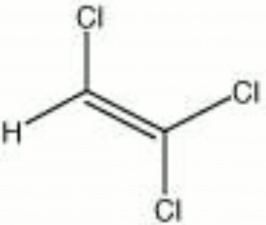
| − | + | [[[SliderGallery rightalign|trichloroethylene.jpg~Chemical structure]]] | |
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
trichloroethene; 1,1,2-trichloroethylene; ethylene trichloride; westrosol; Tri-Clene [DuPont]; Trethylene; Chlorylene; | trichloroethene; 1,1,2-trichloroethylene; ethylene trichloride; westrosol; Tri-Clene [DuPont]; Trethylene; Chlorylene; | ||
| − | [ | + | == Risks == |
| + | |||
| + | * Nonflammable but decomposes with heat to produce toxic fumes. | ||
| + | * Potential carcinogen. | ||
| + | * Toxic by inhalation. | ||
| + | * Usage prohibited in some states. | ||
| + | * Skin contact causes irritation. | ||
| + | * Millipore Sigma: [https://www.emdmillipore.com/US/en/product/msds/MDA_CHEM-100958?ReferrerURL=https%3A%2F%2Fsearch.yahoo.com%2F&bd=1 MSDS] | ||
| − | == | + | ==Physical and Chemical Properties== |
Miscible with organic solvents. Insoluble in water. | Miscible with organic solvents. Insoluble in water. | ||
| Line 22: | Line 29: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | -73 | + | | -73 C |
|- | |- | ||
! scope="row"| Density | ! scope="row"| Density | ||
| − | | 1.456-1.462 | + | | 1.456-1.462 g/ml |
|- | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| Line 34: | Line 41: | ||
|- | |- | ||
! scope="row"| Boiling Point | ! scope="row"| Boiling Point | ||
| − | | 86.7 | + | | 86.7 C |
|} | |} | ||
| − | == | + | == Resources and Citations == |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
* G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 303 | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 303 | ||
Latest revision as of 12:00, 16 June 2022
Description
Colorless liquid with a chloroform odor. Trichloroethylene has been used as a solvent for fats, oils, waxes, resins, Rubber, plastics, paints, and varnishes. It was widely used for degreasing and dry-cleaning, but by 1990 its usage was banned in many states.
Synonyms and Related Terms
trichloroethene; 1,1,2-trichloroethylene; ethylene trichloride; westrosol; Tri-Clene [DuPont]; Trethylene; Chlorylene;
Risks
- Nonflammable but decomposes with heat to produce toxic fumes.
- Potential carcinogen.
- Toxic by inhalation.
- Usage prohibited in some states.
- Skin contact causes irritation.
- Millipore Sigma: MSDS
Physical and Chemical Properties
Miscible with organic solvents. Insoluble in water.
| Composition | CHCl:CCl2 |
|---|---|
| CAS | 79-01-6 |
| Melting Point | -73 C |
| Density | 1.456-1.462 g/ml |
| Molecular Weight | mol. wt.= 131.4 |
| Refractive Index | 1.4735 |
| Boiling Point | 86.7 C |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 303
- Reed Kay, The Painter's Guide To Studio Methods and Materials, Prentice-Hall, Inc., Englewood Cliffs, NJ, 1983
- Ralph Mayer, A Dictionary of Art Terms and Techniques, Harper and Row Publishers, New York, 1969 (also 1945 printing)
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 9769; ref. index=1.4735
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index=1.475