Difference between revisions of "Tartaric acid"
(username removed) |
m (Text replace - "== Authority ==" to "== Sources Checked for Data in Record ==") |
||
| Line 43: | Line 43: | ||
LINK: [http://www.cdc.gov/niosh/ipcsneng/neng0772.html International Chemical Safety Card] | LINK: [http://www.cdc.gov/niosh/ipcsneng/neng0772.html International Chemical Safety Card] | ||
| − | == | + | == Sources Checked for Data in Record == |
* G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 68 | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 68 | ||
Revision as of 18:58, 1 May 2016
Description
Colorless, transparent crystals that occur naturally in wine lees. Tartaric acid is used in baking powders (potassium hydrogen tartrate), leather tanning and effervescent beverages. Tartaric acid acts as a buffering agent and sequestrant. It is used by dyers to print a blue ferric tartrate color and to remove some mordants from solution. Tartaric acid (and tartrates) reacts with ammonio-silver nitrate to produce metallic silver; this reaction is used in photographic developing solutions and for silvering mirrors.
Synonyms and Related Terms
tartar; dihydroxysuccinic acid; L-tartaric acid; racemic acid; uvic acid; Weinsäure (Deut.); Ácido tartárico (Esp., Port.); Wijnsteenzuur (Ned.)
Other Properties
Soluble in water, ethanol, ether, glycerol. Insoluble in chloroform.
Smells of burnt sugar when heated.
pH = 2.2 (0.1 N solution)
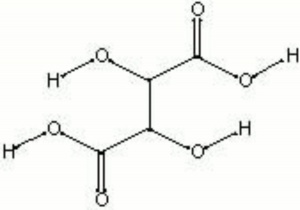
| Composition | HOOC(CH2O)2COOH |
|---|---|
| CAS | 133-37-9 |
| Melting Point | 170 |
| Density | 1.76 |
| Molecular Weight | mol. wt. = 150.1 |
Hazards and Safety
Combustible. Flash point = 210 C
Corrosive. Contact cause irritation and burns.
LINK: International Chemical Safety Card
Sources Checked for Data in Record
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 68
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 9235
- Wikipedia, the free encyclopedia, at http://www.wikipedia.com Comment: http://en.wikipedia.org/wiki/Tartaric_acid (Accessed Sept. 28, 2005)
- S.R.Trotman, E.R. Trotman, Textile Analysis, J.B. Lippincott Company, Philadelphia, 1932
- Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, Technology and Conservation, Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- The American Heritage Dictionary or Encarta, via Microsoft Bookshelf 98, Microsoft Corp., 1998
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: pH = 2.2 (0.1 N solution)
- Website address 1 Comment: www.jetcity.com/~mrjones/chemdesc.htm - photographic chemicals