Difference between revisions of "Zeolite"
m (Text replace - "\[http:\/\/cameo\.mfa\.org\/materials\/fullrecord\.asp\?name=([^\s]+)\s(.*)\]" to "$2") |
|||
| Line 1: | Line 1: | ||
[[File:pc30270chabazite.jpg|thumb|Chabazite]] | [[File:pc30270chabazite.jpg|thumb|Chabazite]] | ||
== Description == | == Description == | ||
| − | + | [[File:ps20510stilbite.jpg|thumb|stilbite]] | |
A group of naturally occurring minerals composed of alkali-hydrated aluminum silicates. Examples are analcite, chabazite, heulandite, [[natrolite|natrolite]], phillipsite, scolecite, stilbite, and thomosonite. Zeolites are soft, fibril minerals with numerous interconnecting voids that can absorb water as well as other liquids and gases. Additionally, zeolites have an ion-exchange capability since its alkaline cations, such as calcium, are mobile and capable of switching with other cations that pass through the cavities. By varying pressure and heat, synthetic zeolite matrices can be prepared from [[silica|silicon dioxide]] and [[aluminum%20oxide|aluminum oxide]] with selected pore sizes and textures ranging from gelatinous to sand-like. Zeolites are used as [[absorbent|absorbents]], [[catalyst|catalysts]], [[desiccant|desiccants]], filters, [[ion%20exchange%20resin|ion exchange resins]], and [[molecular%20sieve|molecular sieves]]. | A group of naturally occurring minerals composed of alkali-hydrated aluminum silicates. Examples are analcite, chabazite, heulandite, [[natrolite|natrolite]], phillipsite, scolecite, stilbite, and thomosonite. Zeolites are soft, fibril minerals with numerous interconnecting voids that can absorb water as well as other liquids and gases. Additionally, zeolites have an ion-exchange capability since its alkaline cations, such as calcium, are mobile and capable of switching with other cations that pass through the cavities. By varying pressure and heat, synthetic zeolite matrices can be prepared from [[silica|silicon dioxide]] and [[aluminum%20oxide|aluminum oxide]] with selected pore sizes and textures ranging from gelatinous to sand-like. Zeolites are used as [[absorbent|absorbents]], [[catalyst|catalysts]], [[desiccant|desiccants]], filters, [[ion%20exchange%20resin|ion exchange resins]], and [[molecular%20sieve|molecular sieves]]. | ||
| − | |||
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| Line 11: | Line 10: | ||
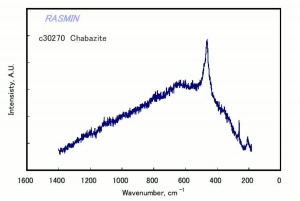
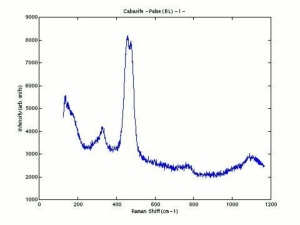
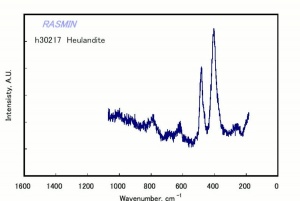
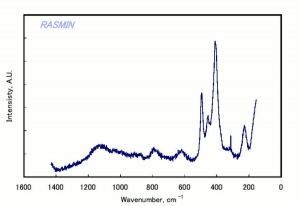
[[[SliderGallery rightalign|chabaziteRS.jpg~Raman|Chabasiteitaly1.jpg~Raman|heulanditeRS.jpg~Raman|stilbiteRS.jpg~Raman|Stilbiteitaly1.jpg~Raman]]] | [[[SliderGallery rightalign|chabaziteRS.jpg~Raman|Chabasiteitaly1.jpg~Raman|heulanditeRS.jpg~Raman|stilbiteRS.jpg~Raman|Stilbiteitaly1.jpg~Raman]]] | ||
| − | == | + | == Physical and Chemical Properties == |
| − | Hexagonal crystal system forming cube-like rhombohedrons. Luster = vitreous. Fracture = uneven. Streak = white. | + | * Hexagonal crystal system forming cube-like rhombohedrons. |
| + | * Luster = vitreous. | ||
| + | * Fracture = uneven. | ||
| + | * Streak = white. | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 24: | Line 26: | ||
|- | |- | ||
! scope="row"| Density | ! scope="row"| Density | ||
| − | | 2.0-2.1 | + | | 2.0-2.1 g/ml |
|- | |- | ||
! scope="row"| Refractive Index | ! scope="row"| Refractive Index | ||
| 1.470-1.494 | | 1.470-1.494 | ||
|} | |} | ||
| − | |||
| − | |||
| − | |||
| − | |||
== Additional Images == | == Additional Images == | ||
| Line 40: | Line 38: | ||
</gallery> | </gallery> | ||
| + | ==Resources and Citations== | ||
| − | + | * A. Dyer, "An Introduction to Zeolite Molecular Sieves', J. Wiley & Sons, London, 1988.° S. Rempel "Zeolite Molecular Traps and their use in Preventive Conservation ", WAAC Newsletter, Vol.18 No. 1 1996 | |
* G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 328 | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 328 | ||
| Line 47: | Line 46: | ||
* Walter C. McCrone, John Gustave Delly, ''The Particle Atlas'', W. McCrone Associates, Chicago, IV, 1972 | * Walter C. McCrone, John Gustave Delly, ''The Particle Atlas'', W. McCrone Associates, Chicago, IV, 1972 | ||
| − | * ''Encyclopedia Britannica'', http://www.britannica.com Comment: "Zeolite." | + | * ''Encyclopedia Britannica'', http://www.britannica.com Comment: "Zeolite." Accessed 19 May 2004. |
* C.W.Chesterman, K.E.Lowe, ''Audubon Society Field Guide to North American Rocks and Minerals'', Alfred A. Knopf, New York, 1979 | * C.W.Chesterman, K.E.Lowe, ''Audubon Society Field Guide to North American Rocks and Minerals'', Alfred A. Knopf, New York, 1979 | ||
| − | * Wikipedia | + | * Wikipedia: http://en.wikipedia.org/wiki/Zeolite (Accessed Sept. 20, 2005) |
[[Category:Materials database]] | [[Category:Materials database]] | ||
Revision as of 09:50, 6 June 2022
Description
A group of naturally occurring minerals composed of alkali-hydrated aluminum silicates. Examples are analcite, chabazite, heulandite, Natrolite, phillipsite, scolecite, stilbite, and thomosonite. Zeolites are soft, fibril minerals with numerous interconnecting voids that can absorb water as well as other liquids and gases. Additionally, zeolites have an ion-exchange capability since its alkaline cations, such as calcium, are mobile and capable of switching with other cations that pass through the cavities. By varying pressure and heat, synthetic zeolite matrices can be prepared from silicon dioxide and Aluminum oxide with selected pore sizes and textures ranging from gelatinous to sand-like. Zeolites are used as absorbents, catalysts, desiccants, filters, ion exchange resins, and molecular sieves.
Synonyms and Related Terms
molecular sieve; Kaken gel; Nikka pellets; Arten gel; MicroChamber; chabazite; analcite; heulandite; natrolite; phillipsite; scolecite; stilbite; thomosonite; seolit (Dan.); Zeolith (Deut.); zeolita (Esp.); zéolithe (Fr.); zeoliet (Ned.); zeólito (Port.)
Physical and Chemical Properties
- Hexagonal crystal system forming cube-like rhombohedrons.
- Luster = vitreous.
- Fracture = uneven.
- Streak = white.
| Composition | Na2O.Al2O3.xSiO2.xH2O |
|---|---|
| Mohs Hardness | 4.0 -5.0 |
| Density | 2.0-2.1 g/ml |
| Refractive Index | 1.470-1.494 |
Additional Images
Resources and Citations
- A. Dyer, "An Introduction to Zeolite Molecular Sieves', J. Wiley & Sons, London, 1988.° S. Rempel "Zeolite Molecular Traps and their use in Preventive Conservation ", WAAC Newsletter, Vol.18 No. 1 1996
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 328
- Walter C. McCrone, John Gustave Delly, The Particle Atlas, W. McCrone Associates, Chicago, IV, 1972
- Encyclopedia Britannica, http://www.britannica.com Comment: "Zeolite." Accessed 19 May 2004.
- C.W.Chesterman, K.E.Lowe, Audubon Society Field Guide to North American Rocks and Minerals, Alfred A. Knopf, New York, 1979
- Wikipedia: http://en.wikipedia.org/wiki/Zeolite (Accessed Sept. 20, 2005)