Difference between revisions of "CI 13065, Metanil Yellow, Acid Yellow 36, LC"
(Created page with " == Synonyms == C.I. 13065; C.I. Acid Yellow 36; C.I. Acid Yellow 36 monosodium salt; C.I. Acid Yellow 36, monosodium salt; C.I. Acid Yellow 36, monosodium salt; 3-(4-Anilino...") |
|||
| Line 10: | Line 10: | ||
Molecular Weight 375.3768 | Molecular Weight 375.3768 | ||
| − | |||
| − | |||
== Analytical instrumentation and procedures == | == Analytical instrumentation and procedures == | ||
Revision as of 11:41, 2 August 2017
Synonyms
C.I. 13065; C.I. Acid Yellow 36; C.I. Acid Yellow 36 monosodium salt; C.I. Acid Yellow 36, monosodium salt; C.I. Acid Yellow 36, monosodium salt; 3-(4-Anilinophenylazo)benzenesulfonic acid sodium salt; 3-[[4-(Phenylamino)phenyl]azo]benzenesulfonic acid monosodium salt; Acid Yellow 36; metanil yellow (C.I. 13065); sodium 3-(p-anilinophenylazo)benzenesulphonate; Acid Yellow 36 (13065); Acid Golden Yellow G; C.I.Acid Yellow 36; [3-(4-anilinophenyl)azophenyl]sulfanyloxyperoxysodium
Molecular Information
Molecular Formula C18H14N3NaO3S
Molecular Weight 375.3768
Analytical instrumentation and procedures
HPLC-DAD-MS analysis was performed with an Agilent 1100 liquid chromatography system consisting of an automatic injector, a gradient pump, a HP series 1100 DAD, and an Agilent series 1100 VL on-line atmospheric pressure ionization electrospray ionization mass spectrometer. Separations were done on a Vydac 214TP52 analytical column (2.1 mm diameterX250 mm; 5-ím particle size). The column was eluted at a flow rate of 0.2 mL/min with a tertiary gradient of water (A),acetonitrile (B), and 1% (v/v) aqueous formic acid (C) with the following elution program: 0 min, 90% A, 5% B, 5% C; 0-55 min, a linear gradient to 35% A, 60% B, 5% C; 55-60 min, a linear gradient elution to 15% A, 80% B, 5% C; 60-62 min, isocratic elution at 15% A, 80% B, 5% C; 62-70 min gradient elution to 90% A, 5% B, 5% C; and reequilibration with the latter solvent for 15 min. The mass spectrometer was run in the positive ion mode.
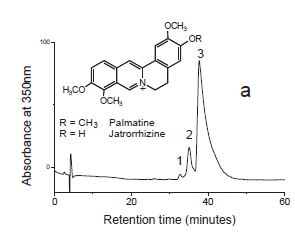
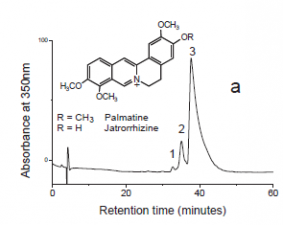
Chromatograms
Fiber sample (~0.5 cm) from Peabody-Essex, Salem was extracted with 1 mL methanol:H2O (v:v=1:1) with 5% formic acid. Then the upper 30 μl of solution was removed for HPLC-DAD-MS analysis (20 μl was injected).
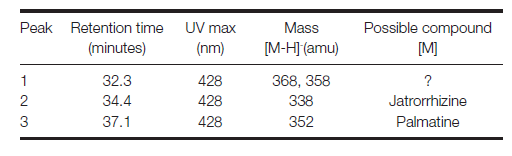
Results
Identified compounds
| Compound | RT (min.) | MW | UV/vis | Other | |
|---|---|---|---|---|---|
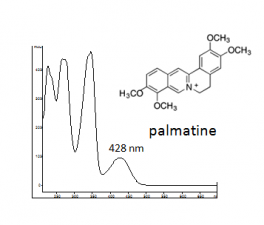
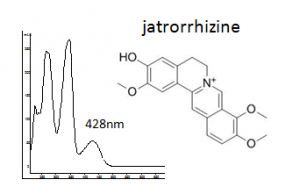
| palmatine | 37.1 | 351 | 348,428 | major component | |
| jatrorrhizine | 34.4 | 337 | 348,428 |