Difference between revisions of "Magnesium stearate"
Jump to navigation
Jump to search
(username removed) |
(username removed) |
||
| Line 1: | Line 1: | ||
== Description == | == Description == | ||
| − | A soft, white powder. Magnesium stearate is used as a dusting powder, a [http://cameo.mfa.org/materials/fullrecord.asp?name=flatting | + | A soft, white powder. Magnesium stearate is used as a dusting powder, a [http://cameo.mfa.org/materials/fullrecord.asp?name=flatting%20agent flatting agent], and a [http://cameo.mfa.org/materials/fullrecord.asp?name=drier drier] in [http://cameo.mfa.org/materials/fullrecord.asp?name=paint paints] and [http://cameo.mfa.org/materials/fullrecord.asp?name=varnish varnishes]. |
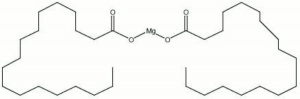
[[[SliderGallery rightalign|magnesium stearate.jpg~Chemical structure]]] | [[[SliderGallery rightalign|magnesium stearate.jpg~Chemical structure]]] | ||
| Line 35: | Line 35: | ||
== Authority == | == Authority == | ||
| − | * | + | * Richard S. Lewis, ''Hawley's Condensed Chemical Dictionary'', Van Nostrand Reinhold, New York, 10th ed., 1993 |
* ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5730 | * ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5730 | ||
Revision as of 06:31, 24 July 2013
Description
A soft, white powder. Magnesium stearate is used as a dusting powder, a flatting agent, and a drier in paints and varnishes.
Other Properties
Insoluble in water and alcohol.
| Composition | Mg(C18H35O2)2 |
|---|---|
| CAS | 557-04-0 |
| Melting Point | 88.5 |
| Density | 1.028 |
| Molecular Weight | mol. wt. = 591.25 |
Hazards and Safety
Nonflammable. Contact may cause irritation.
Fisher Scientific: MSDS
Authority
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5730