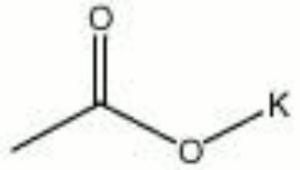
Potassium acetate
Revision as of 12:04, 27 April 2013 by (username removed)
Description
White, deliquescent crystals often used for humidity control. Potassium acetate is used as a dehydrating agent, textile conditioner, an melter ice melter, and as an additive in crystal glass. In a closed environment, a saturated solution of potassium acetate will form an equilibrium at a relative humidity of about 23% (20C).
Synonyms and Related Terms
foliated earth of tartar; acetate of potash
Other Properties
Soluble in water, ethanol. Insoluble in ether.
Deliquescent point at 20C is 23.3 % RH (see salt solutions saturated salt solutions)
| Composition | KC2H3O2 |
|---|---|
| CAS | 127-08-2 |
| Melting Point | 292 |
| Density | 1.57 |
| Molecular Weight | mol. wt. = 98.14 |
Hazards and Safety
Combustible.
International Chemical Safety Card
Authority
- G.S.Brady, G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p.609
- Richard S. Lewis, Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Random House, Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 7764