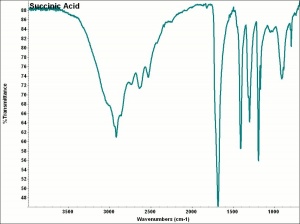
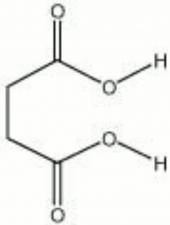
Succinic acid
Jump to navigation
Jump to search
Description
Colorless, odorless crystals that occur naturally in Amber. Succinic acid was first separated from the distillate of amber in 1546 by Agricola. Commercially, succinic acid is used as a Sequestrant and Buffer. It is also used in the manufacture of dyes, lacquers, and photographic solutions.
Synonyms and Related Terms
butanedioic acid; acid of amber; amber acid; ethylenesuccinic acid; BernsteinsSure (Deut.)
Other Properties
Soluble in ethanol, ether. Slightly soluble in water: pH =2.7 (0.1 M solution)
Insoluble in benzene, carbon disulfide, carbon tetrachloride, ligroin.
| Composition | CO2H(CH2)2CO2H |
|---|---|
| CAS | 110-15-6 |
| Melting Point | 185-1.87 |
| Density | 1.55-1.56 |
| Molecular Weight | mol. wt. = 118.1 |
| Boiling Point | 235 |
Hazards and Safety
Contact causes irritation and possible burns.
Mallinckrodt Baker: MSDS
Sources Checked for Data in Record
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 54
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 9037
- Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, Technology and Conservation, Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985