Difference between revisions of "Acrylic acid"
Jump to navigation
Jump to search
(username removed) |
|||
| (3 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
== Description == | == Description == | ||
| − | A colorless, corrosive liquid that polymerizes readily in the presence of oxygen. Acrylic acid was first synthesized in 1843. It is used in the manufacture of [ | + | A colorless, corrosive liquid that polymerizes readily in the presence of oxygen. Acrylic acid was first synthesized in 1843. It is used in the manufacture of [[acrylic%20resin|acrylic resins]]. |
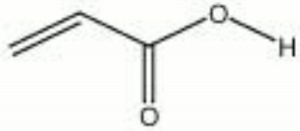
| + | [[[SliderGallery rightalign|acrylic acid.jpg~Chemical structure]]] | ||
| + | == Synonyms and Related Terms == | ||
| − | + | 2-propenoic acid; vinyl formic acid; acroleic acid; propene acid; ácido acrílico (Esp.); ácido 2-propenoico (Esp.) | |
| − | + | == Risks == | |
| − | [ | + | * Irritating to eyes, nose and skin. |
| + | * Toxic by inhalation. | ||
| + | * Combustible. | ||
| + | * ThermoFisher: [https://www.fishersci.com/store/msds?partNumber=AC164250010&productDescription=ACRYLIC+ACID%2C+99%25+1LTACRYLIC&vendorId=VN00032119&countryCode=US&language=en SDS] | ||
| − | == | + | == Physical and Chemical Properties == |
Miscible with water, ethanol and ether. | Miscible with water, ethanol and ether. | ||
| Line 22: | Line 27: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | 14 | + | | 14 C |
|- | |- | ||
! scope="row"| Density | ! scope="row"| Density | ||
| − | | 1.0621 | + | | 1.0621 g/ml|- |
| − | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| mol. wt. = 72.06 | | mol. wt. = 72.06 | ||
|- | |- | ||
! scope="row"| Boiling Point | ! scope="row"| Boiling Point | ||
| − | | 141.0 | + | | 141.0 C |
|} | |} | ||
| − | == | + | ==Resources and Citations== |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | * | + | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 10 |
* ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 132 | * ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 132 | ||
| − | * | + | * Irving Skeist, ''Handbook of Adhesives'', Van Nostrand Reinhold Company, New York, 1977 Comment: p. 528 |
[[Category:Materials database]] | [[Category:Materials database]] | ||
Latest revision as of 10:53, 24 April 2022
Description
A colorless, corrosive liquid that polymerizes readily in the presence of oxygen. Acrylic acid was first synthesized in 1843. It is used in the manufacture of acrylic resins.
Synonyms and Related Terms
2-propenoic acid; vinyl formic acid; acroleic acid; propene acid; ácido acrílico (Esp.); ácido 2-propenoico (Esp.)
Risks
- Irritating to eyes, nose and skin.
- Toxic by inhalation.
- Combustible.
- ThermoFisher: SDS
Physical and Chemical Properties
Miscible with water, ethanol and ether.
| Composition | H2C:CHCOOH | ||
|---|---|---|---|
| CAS | 79-10-7 | ||
| Melting Point | 14 C | ||
| Density | - | Molecular Weight | mol. wt. = 72.06 |
| Boiling Point | 141.0 C |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 10
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 132
- Irving Skeist, Handbook of Adhesives, Van Nostrand Reinhold Company, New York, 1977 Comment: p. 528