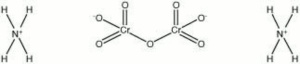
Ammonium dichromate
Jump to navigation
Jump to search
Description
Orange-red prismatic needles. Ammonium dichromate is mixed with Albumin and water to make a light sensitive emulsion for photolithography. It is also used in photography, process engraving, leather tanning, porcelain glazes, textile dyeing, and pyrotechnics.
Synonyms and Related Terms
ammonium bichromate
Risks
- Corrosive. Contact causes burns to skin and membranes.
- Strong Oxidizer. Contact with other materials may cause flames.
- Carcinogenic. Toxic by inhalation and ingestion.
- Fisher Scientific: SDS
Physical and Chemical Properties
Soluble in water and ethanol.
| Composition | (NH4)2Cr2O7 |
|---|---|
| CAS | 7789-09-5 |
| Melting Point | 180 C (dec) |
| Density | 2.155 g/ml |
| Molecular Weight | mol. wt. = 252.07 |
Resources and Citations
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 544