Difference between revisions of "Anthraquinone"
Jump to navigation
Jump to search
(username removed) |
(username removed) |
||
| Line 2: | Line 2: | ||
== Description == | == Description == | ||
| − | Yellow, needle-like crystals that are derived from [http://cameo.mfa.org/materials/fullrecord.asp?name=anthracene anthracene] or [http://cameo.mfa.org/materials/fullrecord.asp?name=phthalic | + | Yellow, needle-like crystals that are derived from [http://cameo.mfa.org/materials/fullrecord.asp?name=anthracene anthracene] or [http://cameo.mfa.org/materials/fullrecord.asp?name=phthalic%20anhydride phthalic anhydride]. Anthraquinone was first sold commercially in 1901. It was used as the starting material in the manufacture of many synthetic dyes, such as [http://cameo.mfa.org/materials/fullrecord.asp?name=alizarin%2C%20synthetic alizarin]. Anthraquinone may be detected by the appearance of a red color on treatment with alkali, [http://cameo.mfa.org/materials/fullrecord.asp?name=zinc zinc] powder, and [http://cameo.mfa.org/materials/fullrecord.asp?name=water water]. |
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| Line 43: | Line 43: | ||
== Authority == | == Authority == | ||
| − | * | + | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 284 |
| − | * | + | * Richard S. Lewis, ''Hawley's Condensed Chemical Dictionary'', Van Nostrand Reinhold, New York, 10th ed., 1993 |
| − | * | + | * Hoechst Celanese Corporation, ''Dictionary of Fiber & Textile Technology'' (older version called Man-made Fiber and Textile Dictionary, 1965), Hoechst Celanese Corporation, Charlotte NC, 1990 |
| − | * | + | * Rosalie Rosso King, ''Textile Identification, Conservation, and Preservation'', Noyes Publications, Park Ridge, NJ, 1985 |
* ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 726 | * ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 726 | ||
| Line 57: | Line 57: | ||
* ''Van Nostrand's Scientific Encyclopedia'', Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976 | * ''Van Nostrand's Scientific Encyclopedia'', Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976 | ||
| − | * | + | * Random House, ''Webster's Encyclopedic Unabridged Dictionary of the English Language'', Grammercy Book, New York, 1997 |
[[Category:Materials database]] | [[Category:Materials database]] | ||
Revision as of 07:25, 24 July 2013
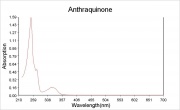
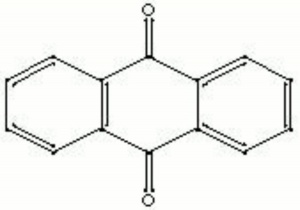
Description
Yellow, needle-like crystals that are derived from anthracene or phthalic anhydride. Anthraquinone was first sold commercially in 1901. It was used as the starting material in the manufacture of many synthetic dyes, such as alizarin. Anthraquinone may be detected by the appearance of a red color on treatment with alkali, zinc powder, and water.
Synonyms and Related Terms
anthroquinone (sp); 9,10-anthracenedione; 9,10-anthraquinone; 9,10-dioxoanthracene; Morkit; Anthradione; Anthrachinon (Deut.); anthraquinon (Fr.); antrachinone (It.)
Other Properties
Soluble in ethanol, ether and acetone. Insoluble in water.
| Composition | C6H4(CO)2C6H4 |
|---|---|
| CAS | 84-65-1 |
| Melting Point | 286 |
| Density | 1.419-1.438 |
| Molecular Weight | mol. wt.=208.05 |
| Boiling Point | 379-381 |
Hazards and Safety
Combustible. Flash point = 185C (365 F). Causes skin irritation.
Fisher Scientific: MSDS
Authority
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 284
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Hoechst Celanese Corporation, Dictionary of Fiber & Textile Technology (older version called Man-made Fiber and Textile Dictionary, 1965), Hoechst Celanese Corporation, Charlotte NC, 1990
- Rosalie Rosso King, Textile Identification, Conservation, and Preservation, Noyes Publications, Park Ridge, NJ, 1985
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 726
- Wikipedia, the free encyclopedia, at http://www.wikipedia.com Comment: http://en.wikipedia.org/wiki/Anthraquinone (Accessed Mar. 20, 2006)
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997