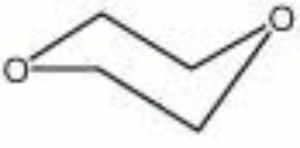
Dioxane
Jump to navigation
Jump to search
Description
A toxic, colorless liquid with a slight ether-like smell. Dioxane is no longer recommended for use. In the past, it was used as a Solvent for Cellulose acetate, Ethyl cellulose, Benzyl cellulose, and other resins, oils, and waxes. It was often found in paint and varnish removers. Dioxane was also used as a Surfactant and Dispersant in textile processing, dyeing, and printing.
Synonyms and Related Terms
diethylene oxide; dioxyethylene ether; 1,4-diethylene dioxide; diethylene ether; 1,4-dioxane
Risks
- Highly toxic by skin contact, inhalation, and ingestion.
- Suspected carcinogen.
- Fumes are highly flammable and explosive. Flash point = 12C (54F)
- ThermoFisher: SDS
Physical and Chemical Properties
Miscible in water and most organic solvents.
| Composition | C4H8O2 |
|---|---|
| CAS | 123-91-1 |
| Melting Point | 11.8 C |
| Density | 1.0329 g/ml |
| Molecular Weight | mol. wt. = 88.1 |
| Refractive Index | 1.420 |
| Boiling Point | 101.1 C |
Resources and Citations
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966
- Hermann Kuhn, Conservation and Restoration of Works of Art and Antiquities, Butterworths, London, 1986
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index = 1.420