Difference between revisions of "Potassium nitrate"
(username removed) |
(username removed) |
||
| Line 2: | Line 2: | ||
== Description == | == Description == | ||
| − | Clear colorless crystals that are slightly [http://cameo.mfa.org/materials/fullrecord.asp?name=hygroscopic hygroscopic]. Potassium nitrate has been used since the 1300s as an ingredient in [http://cameo.mfa.org/materials/fullrecord.asp?name=gunpowder gunpowder]. It was used in [http://cameo.mfa.org/materials/fullrecord.asp?name=gold gold] smelting, [http://cameo.mfa.org/materials/fullrecord.asp?name=glass glass] making, and [http://cameo.mfa.org/materials/fullrecord.asp?name=textile textile] dyeing. In a closed environment, a [http://cameo.mfa.org/materials/fullrecord.asp?name=saturated | + | Clear colorless crystals that are slightly [http://cameo.mfa.org/materials/fullrecord.asp?name=hygroscopic hygroscopic]. Potassium nitrate has been used since the 1300s as an ingredient in [http://cameo.mfa.org/materials/fullrecord.asp?name=gunpowder gunpowder]. It was used in [http://cameo.mfa.org/materials/fullrecord.asp?name=gold gold] smelting, [http://cameo.mfa.org/materials/fullrecord.asp?name=glass glass] making, and [http://cameo.mfa.org/materials/fullrecord.asp?name=textile textile] dyeing. In a closed environment, a [http://cameo.mfa.org/materials/fullrecord.asp?name=saturated%20salt%20solutions saturated salt solution] of potassium nitrate will form an equilibrium at a relative humidity of about 93% (20C). |
[[File:Potassium_nitratekes.jpg|thumb|Powdered possium nitrate]] | [[File:Potassium_nitratekes.jpg|thumb|Powdered possium nitrate]] | ||
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| − | niter; nitre; saltpeter; Bengal saltpeter ; sal prunella; kaliumnitrat (Dan., Deut.); | + | niter; nitre; saltpeter; Bengal saltpeter ; sal prunella; kaliumnitrat (Dan., Deut.); salpêtre (Fr.); nitrato di potassio (It.); kaliumnitraat (Ned.); azotan potasu (Pol.); saletra potasowa (Pol.); |
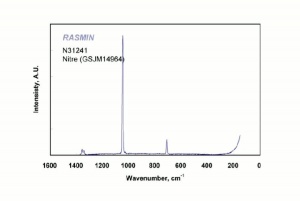
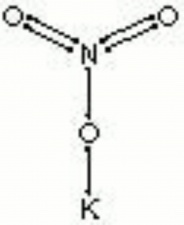
[[[SliderGallery rightalign|nitreRS.jpg~Raman|potassium nitrate.jpg~Chemical structure]]] | [[[SliderGallery rightalign|nitreRS.jpg~Raman|potassium nitrate.jpg~Chemical structure]]] | ||
| Line 15: | Line 15: | ||
Soluble in water, glycerol. Slightly soluble in ethanol. | Soluble in water, glycerol. Slightly soluble in ethanol. | ||
| − | Deliquescent point at 20C is 93.2 % RH (see [http://cameo.mfa.org/materials/fullrecord.asp?name=saturated | + | Deliquescent point at 20C is 93.2 % RH (see [http://cameo.mfa.org/materials/fullrecord.asp?name=saturated%20salt%20solutions saturated salt solutions]) |
{| class="wikitable" | {| class="wikitable" | ||
| Line 46: | Line 46: | ||
== Authority == | == Authority == | ||
| − | * | + | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 632 |
| − | * | + | * Hermann Kuhn, ''Conservation and Restoration of Works of Art and Antiquities'', Butterworths, London, 1986 |
| − | * | + | * Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, ''Technology and Conservation'', Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985 |
| − | * | + | * Michael McCann, ''Artist Beware'', Watson-Guptill Publications, New York City, 1979 |
* Wikipedia, the free encyclopedia, at http://www.wikipedia.com Comment: http://en.wikipedia.org/wiki/Potassium_nitrate (Accessed Nov. 9, 2005) | * Wikipedia, the free encyclopedia, at http://www.wikipedia.com Comment: http://en.wikipedia.org/wiki/Potassium_nitrate (Accessed Nov. 9, 2005) | ||
Revision as of 07:23, 24 July 2013
Description
Clear colorless crystals that are slightly hygroscopic. Potassium nitrate has been used since the 1300s as an ingredient in gunpowder. It was used in gold smelting, glass making, and textile dyeing. In a closed environment, a saturated salt solution of potassium nitrate will form an equilibrium at a relative humidity of about 93% (20C).
Synonyms and Related Terms
niter; nitre; saltpeter; Bengal saltpeter ; sal prunella; kaliumnitrat (Dan., Deut.); salpêtre (Fr.); nitrato di potassio (It.); kaliumnitraat (Ned.); azotan potasu (Pol.); saletra potasowa (Pol.);
Other Properties
Soluble in water, glycerol. Slightly soluble in ethanol.
Deliquescent point at 20C is 93.2 % RH (see saturated salt solutions)
| Composition | KNO3 |
|---|---|
| CAS | 7757-79-1 |
| Melting Point | 333 |
| Density | 2.1062 |
| Molecular Weight | mol. wt. = 101.1 |
| Boiling Point | 400 (dec) |
Hazards and Safety
Dangerous fire and explosion risk when shocked, heated or in contact with organic materials. Strong oxidizing agent.
International Chemical Safety Card
Authority
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 632
- Hermann Kuhn, Conservation and Restoration of Works of Art and Antiquities, Butterworths, London, 1986
- Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, Technology and Conservation, Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- Wikipedia, the free encyclopedia, at http://www.wikipedia.com Comment: http://en.wikipedia.org/wiki/Potassium_nitrate (Accessed Nov. 9, 2005)