Difference between revisions of "Potassium tetraoxalate"
Jump to navigation
Jump to search
(username removed) |
(username removed) |
||
| Line 1: | Line 1: | ||
== Description == | == Description == | ||
| − | White, slightly [http://cameo.mfa.org/materials/fullrecord.asp?name=hygroscopic hygroscopic] crystals. Potassium tetroxalate is used as a water soluble source of [http://cameo.mfa.org/materials/fullrecord.asp?name=oxalic | + | White, slightly [http://cameo.mfa.org/materials/fullrecord.asp?name=hygroscopic hygroscopic] crystals. Potassium tetroxalate is used as a water soluble source of [http://cameo.mfa.org/materials/fullrecord.asp?name=oxalic%20acid oxalic acid] for removing of [http://cameo.mfa.org/materials/fullrecord.asp?name=rust rust] stains and [http://cameo.mfa.org/materials/fullrecord.asp?name=iron%20gall%20ink iron gall inks]. It is also used as a cleaner for metals and wood, as a [http://cameo.mfa.org/materials/fullrecord.asp?name=mordant mordant] in dyeing and as a photographic chemical. |
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| Line 33: | Line 33: | ||
== Authority == | == Authority == | ||
| − | * | + | * Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, ''Technology and Conservation'', Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985 |
| − | * | + | * Hermann Kuhn, ''Conservation and Restoration of Works of Art and Antiquities'', Butterworths, London, 1986 |
| − | * | + | * ''The Merck Index'', Susan Budavari (ed.), Merck Research Labs, Whitehouse Station, NJ, 12th Edition, 1996 Comment: entry 7863 |
[[Category:Materials database]] | [[Category:Materials database]] | ||
Revision as of 07:25, 24 July 2013
Description
White, slightly hygroscopic crystals. Potassium tetroxalate is used as a water soluble source of oxalic acid for removing of rust stains and iron gall inks. It is also used as a cleaner for metals and wood, as a mordant in dyeing and as a photographic chemical.
Synonyms and Related Terms
salts of lemon (incorrect); salt of sorrel; potassium quadoxalate; sal acetosella
Other Properties
Soluble in water. Insoluble in ethanol. Decomposes when heated.
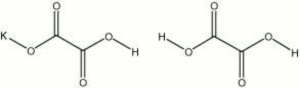
| Composition | KHC2O4 - H2C2O4 |
|---|---|
| CAS | 127-96-8 |
| Molecular Weight | mol. wt. = 254.2 |
Hazards and Safety
Toxic by ingestion.
Mallinckrodt Baker: MSDS
Authority
- Susan E. Schur, Conservation Terminology: A review of Past & Current Nomenclature of Materials, Technology and Conservation, Spring (p.34-39); Summer (p.35-38); Fall (p.25-36), 1985
- Hermann Kuhn, Conservation and Restoration of Works of Art and Antiquities, Butterworths, London, 1986
- The Merck Index, Susan Budavari (ed.), Merck Research Labs, Whitehouse Station, NJ, 12th Edition, 1996 Comment: entry 7863