Difference between revisions of "Linseed oil"
| Line 8: | Line 8: | ||
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| − | + | ''Linum usitatissimum''; flaxseed oil; huile de lin (Fr.); Leinöl (Deut.); aceite de lino (Esp.); aceite de linaza (Esp.); olio di lino (It); lijnzaadolie (Ned.); olej lniany (Pol.); Óleo de linhaça (Port.); linoljan (Sven.); flax seed oil; linum oil | |
Types include: raw; cold-pressed; refined; stand oil; blown oil; bodied oil; boiled oil; sun-refined oil; sun-bleached oil; double boiled oil " | Types include: raw; cold-pressed; refined; stand oil; blown oil; bodied oil; boiled oil; sun-refined oil; sun-bleached oil; double boiled oil " | ||
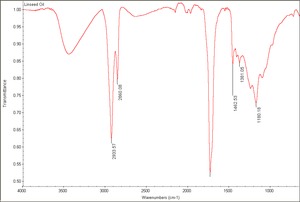
[[[SliderGallery rightalign|Linseed Oil.TIF~FTIR (MFA)]]] | [[[SliderGallery rightalign|Linseed Oil.TIF~FTIR (MFA)]]] | ||
| + | == Risks == | ||
| − | == | + | Contact may cause allergic reaction. Aspiration hazard if swallowed. |
| + | |||
| + | Fisher Scientific: [https://fscimage.fishersci.com/msds/71365.htm MSDS] | ||
| + | == Physical and Chemical == | ||
As a liquid, it is soluble in ether, chloroform, carbon disulfide, ligroin and turpentine. When dry, it is insoluble in most solvents. Saponification number = 190-193. Iodine number = 170-195. Acid number = 1-8. | As a liquid, it is soluble in ether, chloroform, carbon disulfide, ligroin and turpentine. When dry, it is insoluble in most solvents. Saponification number = 190-193. Iodine number = 170-195. Acid number = 1-8. | ||
| Line 33: | Line 37: | ||
|} | |} | ||
| − | == | + | == Resources and Citations == |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | * Jorrit van den Berg, ''Analytical chemical studies on traditional linseed oil paints'', MOLART 2002, available from Archetype Publications, London. | |
| − | + | * M.Serpico, R.White, "Oil, fat and wax" in ''Ancient Egyptian Materials and Technology'', P.Nicholson, I.Shaw (eds.), Cambridge University Press, 2000, p. 390-429. | |
| − | + | * J.S. Mills, R.White, ''The Organic Chemistry of Museum Objects'', Butterworth Heinemann, London, 1994. | |
* ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5335 | * ''The Merck Index'', Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5335 | ||
| − | * ''Encyclopedia Britannica'', http://www.britannica.com Comment: "Linseed." | + | * ''Encyclopedia Britannica'', http://www.britannica.com Comment: "Linseed." Accessed 14 Apr. 2004 . |
* R. J. Gettens, G.L. Stout, ''Painting Materials, A Short Encyclopaedia'', Dover Publications, New York, 1966 Comment: linolenic=36.4-40.3%; linoleic=37.9-45.0%; oleic=13.2-16.0%; stearic and palmitic=4.8-9.0% | * R. J. Gettens, G.L. Stout, ''Painting Materials, A Short Encyclopaedia'', Dover Publications, New York, 1966 Comment: linolenic=36.4-40.3%; linoleic=37.9-45.0%; oleic=13.2-16.0%; stearic and palmitic=4.8-9.0% | ||
Revision as of 11:18, 15 August 2020
Description
A drying oil used in artist paints that is obtained from the seeds of the common flax (Linum usitatissimum) plant. Linseed oil contains the following fatty acids: linolenic (48-60%), oleic (14-24%), linoleic (14-19%), palmitic (6-7%), and stearic (3-6%) (Serpico and White 2000). The drying property is due to the unsaturated bonds in the linoleic and linolenic groups. Linseed oil is the most important and largely used oil for paints and varnishes. It produces a hard, insoluble film when it dries. The yellow-gold color oil is commercially extracted by various methods. The seeds can be crushed in hydraulic or screw-type presses to produce cold-pressed oil. The same process performed on steam-heated seeds produces hot-pressed oil. Cold-pressing is a less efficient manner for extraction, but it produces a higher quality artist paint. Many types of aging, refining, and bleaching procedures have been used to purify the oil and make it dry faster. Linseed oil is used in paints, varnishes, printing inks, synthetic resins, Oilcloth, Linoleum, and soaps.
Synonyms and Related Terms
Linum usitatissimum; flaxseed oil; huile de lin (Fr.); Leinöl (Deut.); aceite de lino (Esp.); aceite de linaza (Esp.); olio di lino (It); lijnzaadolie (Ned.); olej lniany (Pol.); Óleo de linhaça (Port.); linoljan (Sven.); flax seed oil; linum oil
Types include: raw; cold-pressed; refined; stand oil; blown oil; bodied oil; boiled oil; sun-refined oil; sun-bleached oil; double boiled oil "
Risks
Contact may cause allergic reaction. Aspiration hazard if swallowed.
Fisher Scientific: MSDS
Physical and Chemical
As a liquid, it is soluble in ether, chloroform, carbon disulfide, ligroin and turpentine. When dry, it is insoluble in most solvents. Saponification number = 190-193. Iodine number = 170-195. Acid number = 1-8.
| CAS | 8001-26-1 |
|---|---|
| Melting Point | -24.0 |
| Density | 0.921-0.936 |
| Refractive Index | 1.48 -1.49 |
Resources and Citations
- Jorrit van den Berg, Analytical chemical studies on traditional linseed oil paints, MOLART 2002, available from Archetype Publications, London.
- M.Serpico, R.White, "Oil, fat and wax" in Ancient Egyptian Materials and Technology, P.Nicholson, I.Shaw (eds.), Cambridge University Press, 2000, p. 390-429.
- J.S. Mills, R.White, The Organic Chemistry of Museum Objects, Butterworth Heinemann, London, 1994.
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 5335
- Encyclopedia Britannica, http://www.britannica.com Comment: "Linseed." Accessed 14 Apr. 2004 .
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966 Comment: linolenic=36.4-40.3%; linoleic=37.9-45.0%; oleic=13.2-16.0%; stearic and palmitic=4.8-9.0%
- Reed Kay, The Painter's Guide To Studio Methods and Materials, Prentice-Hall, Inc., Englewood Cliffs, NJ, 1983
- Ralph Mayer, A Dictionary of Art Terms and Techniques, Harper and Row Publishers, New York, 1969 (also 1945 printing)
- Robert Feller, Nathan Stolow, Elizabeth Jones, On Picture Varnishes and Their Solvents, National Gallery of Art, Washington DC, 1985
- Matt Roberts, Don Etherington, Bookbinding and the Conservation of Books: a Dictionary of Descriptive Terminology, U.S. Government Printing Office, Washington DC, 1982
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Pam Hatchfield, Pollutants in the Museum Environment, Archetype Press, London, 2002
- Art and Architecture Thesaurus Online, http://www.getty.edu/research/tools/vocabulary/aat/, J. Paul Getty Trust, Los Angeles, 2000
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: melting point = -24.0, density=0.938, ref. index = 1.4782, iodine value=1785.7, saponification value = 190.3