Difference between revisions of "Abietic acid"
Jump to navigation
Jump to search
| (2 intermediate revisions by 2 users not shown) | |||
| Line 2: | Line 2: | ||
Yellowish, semi-crystalline powder. Abietic acid is the primary resin acid component of the solid portion ([[rosin|rosin]]) of resinous exudations obtained from coniferous trees after the volatiles ([[turpentine%20%28oil%29|turpentine]]) are removed. Esters of abietic acid, such as methyl abietate, are used in lacquers and varnishes. Resinates of heavy metals, such as lead abietate, are used as driers in oil paints and varnishes. [[Copper%20resinate|Copper resinate]], formed from mixing a copper salt with rosin, is used as a transparent green pigment. | Yellowish, semi-crystalline powder. Abietic acid is the primary resin acid component of the solid portion ([[rosin|rosin]]) of resinous exudations obtained from coniferous trees after the volatiles ([[turpentine%20%28oil%29|turpentine]]) are removed. Esters of abietic acid, such as methyl abietate, are used in lacquers and varnishes. Resinates of heavy metals, such as lead abietate, are used as driers in oil paints and varnishes. [[Copper%20resinate|Copper resinate]], formed from mixing a copper salt with rosin, is used as a transparent green pigment. | ||
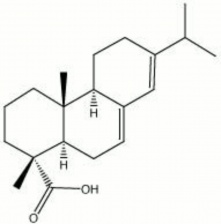
| − | + | [[[SliderGallery rightalign|abietic acid.jpg~Chemical structure]]]== Synonyms and Related Terms == | |
| − | == Synonyms and Related Terms == | ||
abietinic acid; sylvic acid; 13-isopropylpodocapra-7-13-dien-15-oicacid | abietinic acid; sylvic acid; 13-isopropylpodocapra-7-13-dien-15-oicacid | ||
| − | + | == Risks == | |
| − | + | Combustible. Known contact allergen. | |
| − | + | Cayman Chemicals: [https://cdn.caymanchem.com/cdn/msds/24927m.pdf SDS] | |
| − | HLB=8.2 cmc=2 pKa1=6.7 | + | == Physical and Chemical Properties == |
| + | |||
| + | * Soluble in ethanol, acetone, ether, chloroform and benzene. | ||
| + | * Insoluble in water. | ||
| + | * HLB=8.2 | ||
| + | * cmc=2 | ||
| + | * pKa1=6.7 | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 24: | Line 29: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | 172-175 | + | | 172-175 C |
|- | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| Line 33: | Line 38: | ||
|} | |} | ||
| − | == | + | ==Resources and Citations== |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
* Richard C. Wolbers, Nanette T. Sterman, Chris Stavroudis, ''Notes for Workshop on New Methods in the Cleaning of Paintings'', J.Paul Getty Trust, Los Angeles, 1990 | * Richard C. Wolbers, Nanette T. Sterman, Chris Stavroudis, ''Notes for Workshop on New Methods in the Cleaning of Paintings'', J.Paul Getty Trust, Los Angeles, 1990 | ||
| Line 50: | Line 49: | ||
* Teri Hensick, contributed information, 1998 | * Teri Hensick, contributed information, 1998 | ||
| − | |||
| − | |||
| − | |||
[[Category:Materials database]] | [[Category:Materials database]] | ||
Latest revision as of 12:21, 18 April 2022
Description
Yellowish, semi-crystalline powder. Abietic acid is the primary resin acid component of the solid portion (Rosin) of resinous exudations obtained from coniferous trees after the volatiles (turpentine) are removed. Esters of abietic acid, such as methyl abietate, are used in lacquers and varnishes. Resinates of heavy metals, such as lead abietate, are used as driers in oil paints and varnishes. Copper resinate, formed from mixing a copper salt with rosin, is used as a transparent green pigment.
Synonyms and Related Terms
abietinic acid; sylvic acid; 13-isopropylpodocapra-7-13-dien-15-oicacid
Risks
Combustible. Known contact allergen.
Cayman Chemicals: SDS
Physical and Chemical Properties
- Soluble in ethanol, acetone, ether, chloroform and benzene.
- Insoluble in water.
- HLB=8.2
- cmc=2
- pKa1=6.7
| Composition | C19H29COOH |
|---|---|
| CAS | 514-10-3 |
| Melting Point | 172-175 C |
| Molecular Weight | mol. wt. = 302.45 |
| Refractive Index | optical rotation = -106 |
Resources and Citations
- Richard C. Wolbers, Nanette T. Sterman, Chris Stavroudis, Notes for Workshop on New Methods in the Cleaning of Paintings, J.Paul Getty Trust, Los Angeles, 1990
- The Merck Index, Susan Budavari (ed.), Merck Research Labs, Whitehouse Station, NJ, 12th Edition, 1996 Comment: entry 1
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 615
- Teri Hensick, contributed information, 1998