Difference between revisions of "Zircon"
| Line 1: | Line 1: | ||
| − | [[File:1998.569-SC8319.jpg|thumb| | + | [[File:1998.569-SC8319.jpg|thumb|Jeweled brooch<br>MFA# 1998.569]] |
== Description == | == Description == | ||
| − | + | [[File:pz30324zircon2.jpg|thumb|Zircon]] | |
A natural mineral ore of zirconium silicate primarily used to produce [[zirconium]]. The zircon tetragonal prism crystals are found on beaches or in river placer deposits in association with [[silica]] containing minerals. The hard, refractory, but brittle crystals come in a wide range of colors: yellow, green, blue, red to brown. Clear zircon crystals, sold as Matura diamonds, make very brilliant gemstones because of their high refractive index. Clear yellow to red zircon, called [[hyacinth]] or jacinth, is also used as a gem. Ancient gems of hessonite garnets were also called jacinth and hyacinth which led to some being incorrectly labeled zircon (Ogden 1982). Zircons from Sri Lanka have been used for 2000 years. Large deposits are found in Australia. Other sources are Brazil, India, Pakistan, Thailand, New Zealand, Myanmar (formerly Burma), Western Africa, Norway (Seiland), Russia, Canada (Ontario) and the U.S. (Florida, Colorado, South Carolina, New Jersey). | A natural mineral ore of zirconium silicate primarily used to produce [[zirconium]]. The zircon tetragonal prism crystals are found on beaches or in river placer deposits in association with [[silica]] containing minerals. The hard, refractory, but brittle crystals come in a wide range of colors: yellow, green, blue, red to brown. Clear zircon crystals, sold as Matura diamonds, make very brilliant gemstones because of their high refractive index. Clear yellow to red zircon, called [[hyacinth]] or jacinth, is also used as a gem. Ancient gems of hessonite garnets were also called jacinth and hyacinth which led to some being incorrectly labeled zircon (Ogden 1982). Zircons from Sri Lanka have been used for 2000 years. Large deposits are found in Australia. Other sources are Brazil, India, Pakistan, Thailand, New Zealand, Myanmar (formerly Burma), Western Africa, Norway (Seiland), Russia, Canada (Ontario) and the U.S. (Florida, Colorado, South Carolina, New Jersey). | ||
| − | + | [[File:Zirconemr1.jpg|thumb|Zircon sand]] | |
| − | [[File: | ||
| − | |||
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
| Line 12: | Line 10: | ||
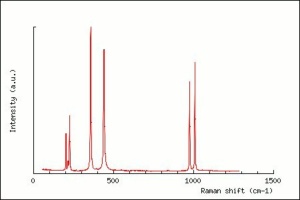
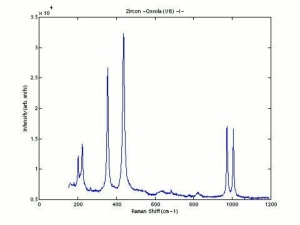
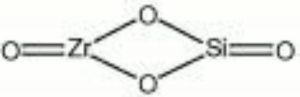
[[[SliderGallery rightalign|zirconlyon.jpg~Raman Lyon|Zirconitaly1.jpg~Raman Parma|Jacinth yellow zircon.jpg~Raman Jacinth MFA|zircon.jpg~Chemical structure]]] | [[[SliderGallery rightalign|zirconlyon.jpg~Raman Lyon|Zirconitaly1.jpg~Raman Parma|Jacinth yellow zircon.jpg~Raman Jacinth MFA|zircon.jpg~Chemical structure]]] | ||
| − | == | + | == Risks == |
| − | |||
| − | |||
| − | + | * Contact and inhalation of zirconium compounds may cause nodules under the skin and in the lungs. | |
| + | * Fisher Scientific: [https://fscimage.fishersci.com/msds/33934.htm MSDS] | ||
| − | + | == Physical and Chemical Properties == | |
| − | Pleochroic. Fluorescence = some show dull yellow color; some may phosphoresce. | + | * Insoluble in acids. |
| + | * Tetragonal system with square prismatic crystals. | ||
| + | * Fracture = uneven. | ||
| + | * Luster = adamantine to vitreous. | ||
| + | * Streak = colorless. | ||
| + | * Pleochroic. | ||
| + | * Fluorescence = some show dull yellow color; some may phosphoresce. | ||
Heating brown zircon crystals produces strong colors (blue, green, red, etc.) that fade slowly with time or with UV exposure. | Heating brown zircon crystals produces strong colors (blue, green, red, etc.) that fade slowly with time or with UV exposure. | ||
| Line 36: | Line 39: | ||
|- | |- | ||
! scope="row"| Melting Point | ! scope="row"| Melting Point | ||
| − | | 2550 | + | | 2550 C |
|- | |- | ||
! scope="row"| Density | ! scope="row"| Density | ||
| − | | 4.6-4.7 | + | | 4.6-4.7 g/ml |
|- | |- | ||
! scope="row"| Molecular Weight | ! scope="row"| Molecular Weight | ||
| Line 47: | Line 50: | ||
| 1.94; 1.98 | | 1.94; 1.98 | ||
|} | |} | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
== Comparisons == | == Comparisons == | ||
| Line 64: | Line 57: | ||
[[media:download_file_441.pdf|Natural and Simulated Diamonds]] | [[media:download_file_441.pdf|Natural and Simulated Diamonds]] | ||
| − | + | ==Resources and Citations== | |
| − | |||
| − | == | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
* Jack Odgen, ''Jewellery of the Ancient World'', Rizzoli International Publications Inc., New York City, 1982 | * Jack Odgen, ''Jewellery of the Ancient World'', Rizzoli International Publications Inc., New York City, 1982 | ||
| Line 79: | Line 63: | ||
* R.F.Symmes, T.T.Harding, Paul Taylor, ''Rocks, Fossils and Gems'', DK Publishing, Inc., New York City, 1997 | * R.F.Symmes, T.T.Harding, Paul Taylor, ''Rocks, Fossils and Gems'', DK Publishing, Inc., New York City, 1997 | ||
| − | * ''Encyclopedia Britannica'', http://www.britannica.com Comment: "zircon" | + | * ''Encyclopedia Britannica'', http://www.britannica.com Comment: "zircon" [Accessed December 11, 2001]. (color photo and tech Info) |
| − | * Website | + | * Website: http://www.geo.utexas.edu/courses/347k/redesign/gem_notes/Zircon/zircon_triple.htm (fluorescence information) |
* C.W.Chesterman, K.E.Lowe, ''Audubon Society Field Guide to North American Rocks and Minerals'', Alfred A. Knopf, New York, 1979 | * C.W.Chesterman, K.E.Lowe, ''Audubon Society Field Guide to North American Rocks and Minerals'', Alfred A. Knopf, New York, 1979 | ||
| − | * Wikipedia | + | * Wikipedia: http://en.wikipedia.org/wiki/Zircon (Accessed Sept. 20, 2005) |
* G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 890 | * G.S.Brady, ''Materials Handbook'', McGraw-Hill Book Co., New York, 1971 Comment: p. 890 | ||
Revision as of 13:07, 2 June 2022
Description
A natural mineral ore of zirconium silicate primarily used to produce Zirconium. The zircon tetragonal prism crystals are found on beaches or in river placer deposits in association with Silica containing minerals. The hard, refractory, but brittle crystals come in a wide range of colors: yellow, green, blue, red to brown. Clear zircon crystals, sold as Matura diamonds, make very brilliant gemstones because of their high refractive index. Clear yellow to red zircon, called Hyacinth or jacinth, is also used as a gem. Ancient gems of hessonite garnets were also called jacinth and hyacinth which led to some being incorrectly labeled zircon (Ogden 1982). Zircons from Sri Lanka have been used for 2000 years. Large deposits are found in Australia. Other sources are Brazil, India, Pakistan, Thailand, New Zealand, Myanmar (formerly Burma), Western Africa, Norway (Seiland), Russia, Canada (Ontario) and the U.S. (Florida, Colorado, South Carolina, New Jersey).
Synonyms and Related Terms
zirconium silicate; zirkelite; zirconite; zircon sand; Matura diamonds; jacinth; hyacinth; jargoon; jargon; Zircopax [Titanium Alloy Mfg.]; Superpax; Ultrox [M&T Chemical]; cubic zirconia; Zirkon (Deut.); zircón (Esp.); circón (Esp.); zircon (Fr.); zirkoon (Ned.); cyrkon (Pol.); zircão (Port.)
Risks
- Contact and inhalation of zirconium compounds may cause nodules under the skin and in the lungs.
- Fisher Scientific: MSDS
Physical and Chemical Properties
- Insoluble in acids.
- Tetragonal system with square prismatic crystals.
- Fracture = uneven.
- Luster = adamantine to vitreous.
- Streak = colorless.
- Pleochroic.
- Fluorescence = some show dull yellow color; some may phosphoresce.
Heating brown zircon crystals produces strong colors (blue, green, red, etc.) that fade slowly with time or with UV exposure.
| Composition | ZrSiO4 |
|---|---|
| CAS | 14940-68-2 |
| Mohs Hardness | 6.0 - 7.5 |
| Melting Point | 2550 C |
| Density | 4.6-4.7 g/ml |
| Molecular Weight | mol. wt. = 183.31 |
| Refractive Index | 1.94; 1.98 |
Comparisons
Properties of Common Gemstones
Natural and Simulated Diamonds
Resources and Citations
- Jack Odgen, Jewellery of the Ancient World, Rizzoli International Publications Inc., New York City, 1982
- R.F.Symmes, T.T.Harding, Paul Taylor, Rocks, Fossils and Gems, DK Publishing, Inc., New York City, 1997
- Encyclopedia Britannica, http://www.britannica.com Comment: "zircon" [Accessed December 11, 2001]. (color photo and tech Info)
- Website: http://www.geo.utexas.edu/courses/347k/redesign/gem_notes/Zircon/zircon_triple.htm (fluorescence information)
- C.W.Chesterman, K.E.Lowe, Audubon Society Field Guide to North American Rocks and Minerals, Alfred A. Knopf, New York, 1979
- Wikipedia: http://en.wikipedia.org/wiki/Zircon (Accessed Sept. 20, 2005)
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 890
- The Merck Index, Susan Budavari (ed.), Merck Research Labs, Whitehouse Station, NJ, 12th Edition, 1996 Comment: entry 9986
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979