Difference between revisions of "Sodium sulfite"
Jump to navigation
Jump to search
m (Text replace - "== Authority ==" to "== Sources Checked for Data in Record ==") |
m (Text replace - "\[http:\/\/cameo\.mfa\.org\/materials\/fullrecord\.asp\?name=([^\s]+)\s(.*)\]" to "$2") |
||
| Line 1: | Line 1: | ||
== Description == | == Description == | ||
| − | White, crystalline powder. Sodium sulfite is primarily used as a reducing agent in photographic fixers and as a replacement for 'hypo' ([ | + | White, crystalline powder. Sodium sulfite is primarily used as a reducing agent in photographic fixers and as a replacement for 'hypo' ([[sodium%20thiosulfate|sodium thiosulfate]]). It is also used for bleaching [[wool|wool]], [[straw|straw]], and [[silk|silk]]. Sodium sulfite acts as an [[antichlor%20agent|antichlor]] for the removal of [[chlorine|chlorine]] in bleached [[textile|textiles]] and [[paper|paper]], but it can leave a residual [[sulfur|sulfur]] odor. It is also used for silvering [[glass|glass]] and preserving food. |
== Synonyms and Related Terms == | == Synonyms and Related Terms == | ||
Revision as of 11:19, 10 May 2016
Description
White, crystalline powder. Sodium sulfite is primarily used as a reducing agent in photographic fixers and as a replacement for 'hypo' (Sodium thiosulfate). It is also used for bleaching Wool, Straw, and Silk. Sodium sulfite acts as an antichlor for the removal of Chlorine in bleached textiles and Paper, but it can leave a residual Sulfur odor. It is also used for silvering Glass and preserving food.
Synonyms and Related Terms
sodium sulphite; disodium salt of sulfurous acid; disodium sulfite
Other Properties
Soluble in water, glycerol. Solutions smell sulfurous.
Insoluble in ethanol.
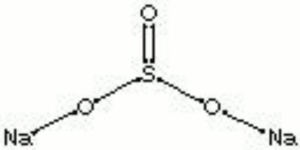
| Composition | NaSO3 |
|---|---|
| CAS | 7757-83-7 |
| Melting Point | 600 (dec) |
| Density | 2.633 |
| Molecular Weight | mol. wt. = 126.06 |
| Refractive Index | 1.565, 1.515 |
Hazards and Safety
Emits toxic fumes when heated.
Inhalation of powder may cause asthmatic reactions.
LINK: International Chemical Safety Card
Sources Checked for Data in Record
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Book and Paper Group, Paper Conservation Catalog, AIC, 1984, 1989
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 8831
- Website address 1 Comment: Photographic chemical at www.jetcity.com/~mrjones/chemdesc.htm
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index= 1.565, 1.515
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- The American Heritage Dictionary or Encarta, via Microsoft Bookshelf 98, Microsoft Corp., 1998