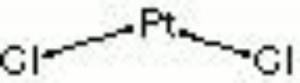Platinum chloride
Jump to navigation
Jump to search
Description
Brown powder. Platinum chloride is formed in conjunction with Chloroplatinic acid by dissolving Platinum in Aqua regia. Although separate compounds, the name platinum chloride has been used commercially for both materials. Chloroplatinic acid is the most commonly used of the two materials. It is used to make platinum photographic prints, indelible ink, and mirrors. It is also used for platinizing glass, porcelain, and pumice stone.
Synonyms and Related Terms
platinum tetrachloride; platinic chloride; platinum (III) chloride
Risks
- Inhalation and skin contact causes irritation and allergic reactions.
- Fisher Scientific: MSDS
Physical and Chemical Properties
Soluble in water, ethanol.
| Composition | PtCl4 |
|---|---|
| CAS | 10025-65-7 |
| Molecular Weight | mol. wt. = 266.0 |
Resources and Citations
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979