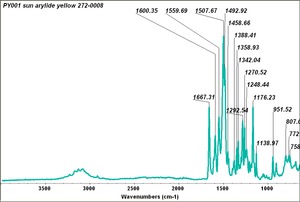
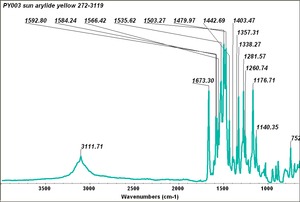
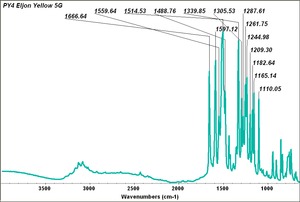
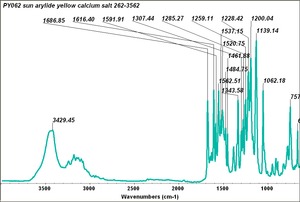
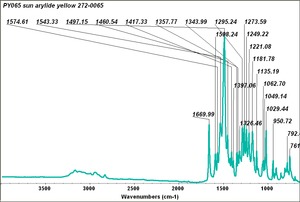
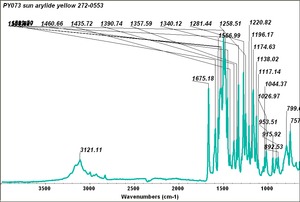
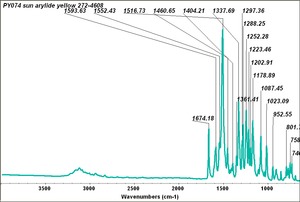
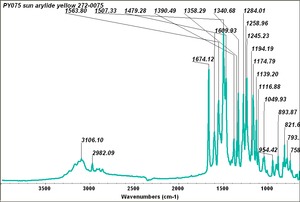
Arylide yellow
Description
A name used for synthetic monoazo yellow colorants. The first arylide yellow (PY 01) was synthesized from aniline-based diazonium salts and acetoacetarylide in 1909 by Hermann Wagner in Germany. Initially they were sold commercially as Hansa yellow by Hoechst AG. Arylide yellows have good tinting strength, opacity and solvent resistance. Because of their good lightfastness they were (and still are) primarily used in printing inks, plastic, rubbers, as well as architectural and artists paints. Monoarylide yellows have better lightfastness than diarylides. The colors began to be commercially available around 1925 and became more widely used mid-century as a replacement for the toxic cadmium yellow. There have been some arylide oranges (PO1, PO6) and reds (PR211), but they are no used as artist colorants.
Synonyms and Related Terms
monoarylide; azo dye; monoazo dye; Hansa yellow; colorante azoico (Esp.); colorante azoico (It.)
Arylide Dye Comparisons
Note: Most information in this table is from Artist's pigment.org. Their pages have more compounds as well as Health & Safety info.
| Pigment number | CI number | Chemical composition | Discovery or Patent; Production |
Some commercial names | Description |
|---|---|---|---|---|---|
| PY01 | 11680 | C17H16N4O4 | 1909; 1910 | Hansa Yellow G; Primary yellow; Fast Yellow G | greenish yellow, lower color strength |
| PY02 | 11730 | C18H17ClN4O4 | 1911; 1920 | Hansa Yellow GR | more reddish shade than PY1 |
| PY03 | 11710 | C16H12Cl2N4O4 | 1910; 1912 | Hansa Yellow 10G; Hansa Yellow light; Studio Yellow; Monoazo Yellow Lemon | green shade |
| PY04 | 11665 | C16H14N4O4 | 1909 | Hansa Yellow 10G; | |
| PY05 | 11660 | C16H14N4O4 | 1909; 1928 | Hansagelb5G; | |
| PY06 | 11670 | C16H13ClN4O4 | 1909; 1928 | Hansa Yellow 3G; | orangish |
| PY10 | 12710 | C16H12Cl2N4O | xx; 1928 | Hansa Yellow R | |
| PY49 | 11765 | C19H19Cl2N3O4 | Monolite Fast Yellow | ||
| PY60 | 12705 | C16H13ClN4O | xx; 1928 | Hansa Yellow 4R3D-1750; | |
| PY62 | 13940 | Ca; C17H16N4O7S | Azo Yellow 62; HP Yellow | ||
| PY65 | 11740 | C18H18N4O6 | 1936; xx | Hansa Yellow 3RN; Hansa Yellow Deep; Permanent Yellow Deep | reddish shade |
| PY73 | 11738 | C17H15ClN4O5 | 1957; xx | Arylide Yellow GX; Hansa Brilliant Yellow; Hansa Yellow Medium | mid yellow |
| PY74 | 11741 | C18H18N4O6 | 1958; 1961 | Arylide Yellow GX; Arylide Yellow 5GX; Hansa Brilliant Yellow | green tinge, high color strength |
| PY75 | 11770 | C18H17ClN4O5 | Arylide Yellow; Hansa Yellow | redder than PY74 |
Physical and Chemical Properties
- Most arylide dyes are soluble in organic solvents and some may discolor in solvents.
- Resistant to water, oil, acids and bases.
- Melting Point = 150 (dec)
Risks
- May bleed in paints.
- Decomposes at temperatures over 150 C.
- Potential carcinogen.
Resources and Citations
- ColourLex: Arylide Yellow
- B. Berrie, S.Q. Lomax, 'Azo Pigments: Their History, Synthesis, Properties and Use in Artists' Materials', Studies in the History of Art, National Gallery of Art, Washington DC, No. 57, 1997
- Artist Pigments.org: Pigment Yellow Note: see individual sheets for each pigment number
- Handprint.com: Monoazo
- Wikipedia: Arylide yellow Accessed March 2026
- CHSOS: Spectra (Reflectance, XRF, Raman, FTIR) for Arylide yellow 5GX