Cadmium yellow
Description
A permanent, yellow pigment composed of cadmium sulfide. Cadmium yellows were synthetically prepared in Germany by Friedrich Strohmeyer in 1817. The bright yellow pigments slowly began to be used as artist paints in the mid 1840s, gaining in popularity in the early 20th century. Variations in particle size and chemical composition produce as range of colors from light yellow to orange. In the 1920s, the cadmium pigments were co-precipitated with barium sulfate to form the cheaper cadmium lithopone (cadmopone) pigments. Another variation of cadmium yellow was a solid solution of cadmium sulfide with zinc sulfide (known as PY 35). Cadmium sulfide also occurs naturally in minor amounts in the mineral greenockite.
Synonyms and Related Terms
cadmium sulfide; Pigment Yellow 37; CI 77191; Kadmiumgelb (Deut.); jaune de cadmium (Fr.); cadmium sulphide (Br.); giallo di cadmio (It.); amarillo de cadmio (Esp.); kitrino toy kadmioy (Gr.); cadmiumgeel (Ned.); amarelo de cádmio (Port.); cadmium lithopone; cadmopone; Aurora yellow; daffodil; radiant yellow; cadmia; Orient yellow; jaune brilliant; Cadmolith
Applications
- Paint pigments
- Coloring glass
Risks
- Highly toxic by inhalation such as dust in colored chalk, vapors from welding or spray paint application
- Ingestion has always been unconcerning since it is very insoluble
- Carcinogen.
- M.Graham and Co. Watercolor: SDS
Physical and Chemical Properties
- Cubic or hexagonal crystals.
- Soluble in concentrated mineral acids with the evolution of H2S. Insoluble in water.
- May fluoresce red.
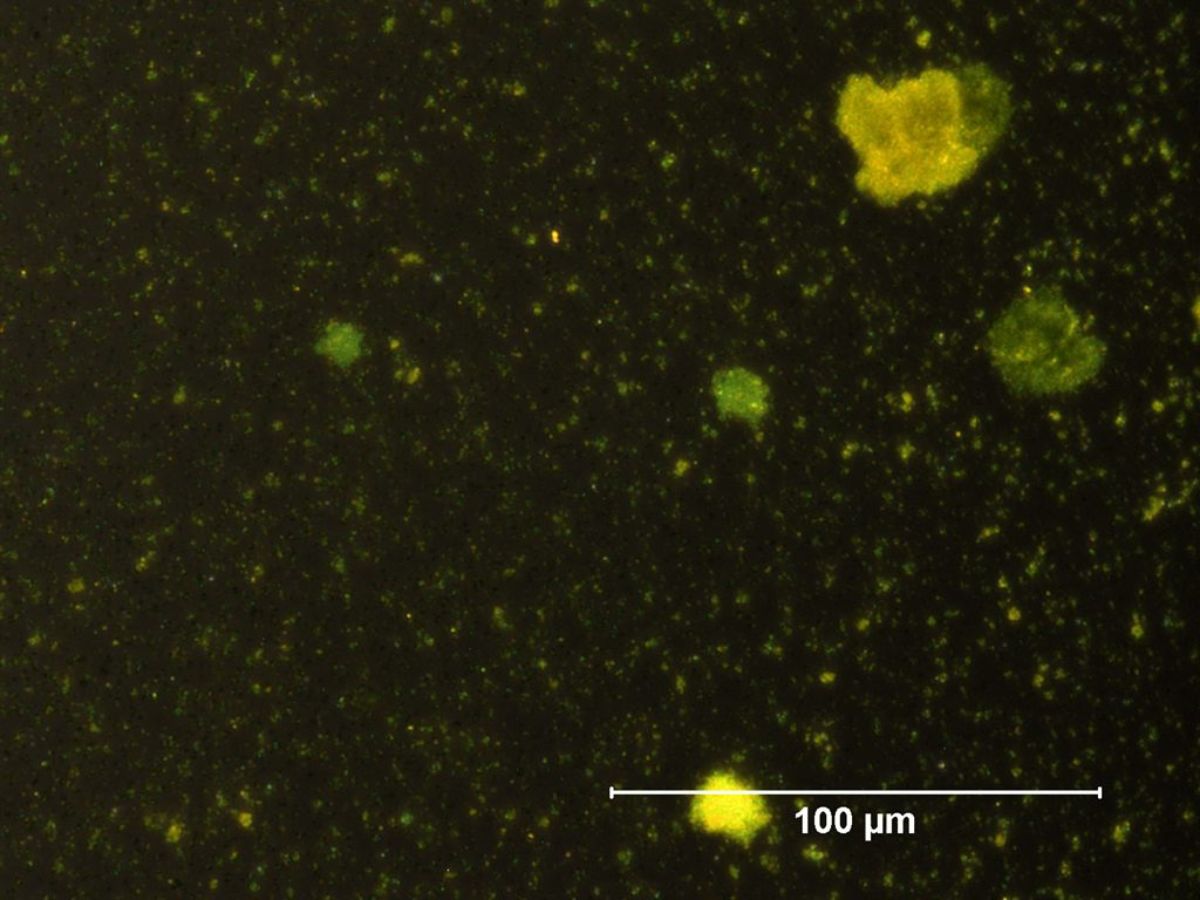
- The tiny yellow particles (about 1 micrometer) have a high refractive index.
- Composition = CdS (mol wt. = 144.48 g/mol)
- Density = 4.35 g/ml
- Refractive Index = e=2.506, w=2.529
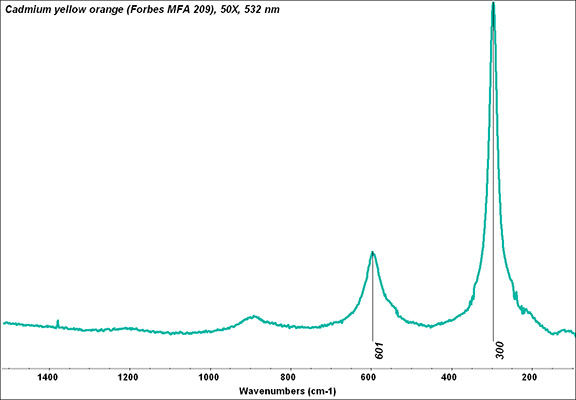
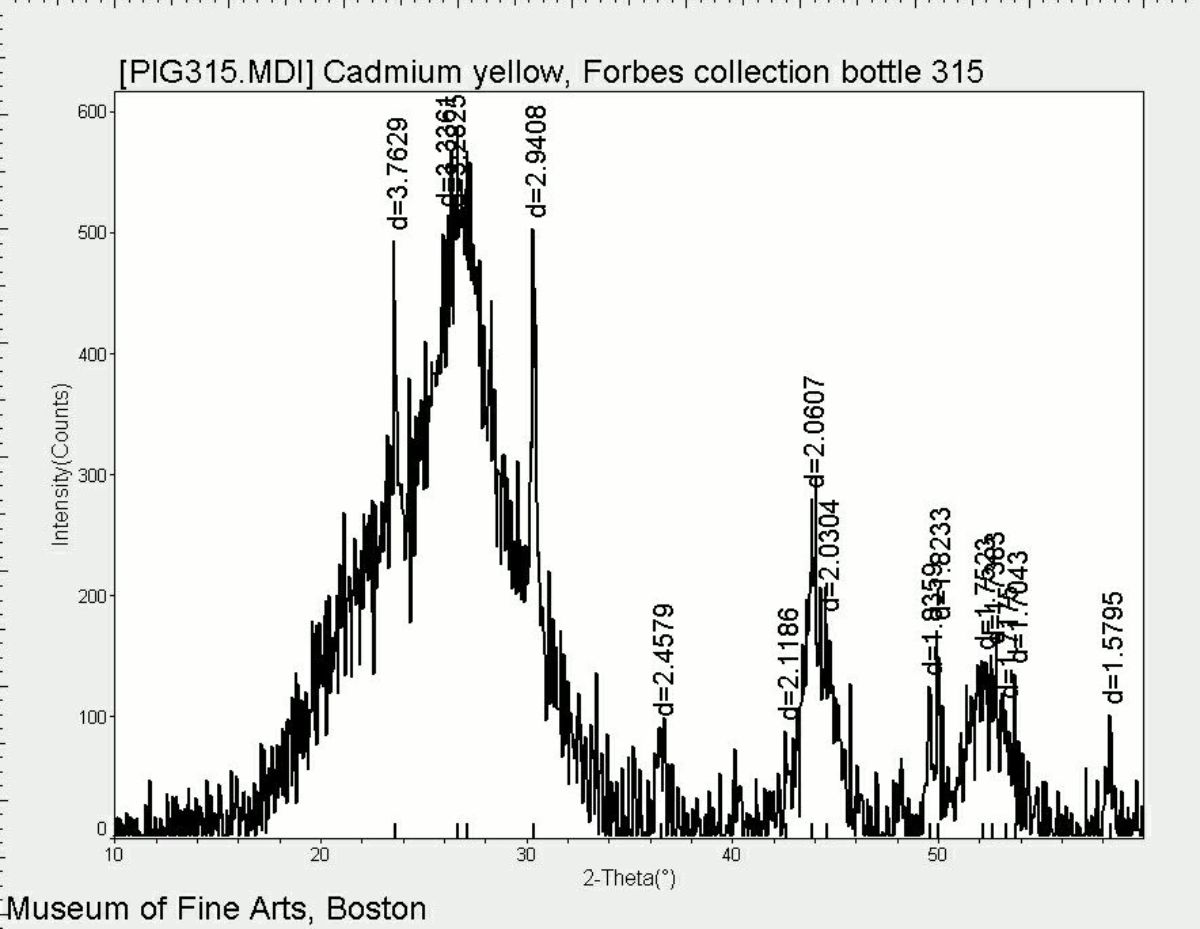
Additional Images
Resources and Citations
- I. Fiedler, M. Bayard, "Cadmium yellows, oranges and reds", Artists Pigments, Volume 1, R. Feller (ed.), Cambridge University Press: Cambridge, 1986.
- Nicholas Eastaugh, Valentine Walsh, Tracey Chaplin, Ruth Siddall, Pigment Compendium, Elsevier Butterworth-Heinemann, Oxford, 2004.
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966 Comment: density = 4.35 and ref. index = 2.35-2.48
- Ralph Mayer, A Dictionary of Art Terms and Techniques, Harper and Row Publishers, New York, 1969 (also 1945 printing)
- Pigments Through the Ages: http://webexhibits.org/pigments/indiv/technical/cdyellow.html - e=2.506, w=2.529
- Thomas B. Brill, Light Its Interaction with Art and Antiquities, Plenum Press, New York City, 1980
- The Dictionary of Art, Grove's Dictionaries Inc., New York, 1996 Comment: 'Pigments'
- Art and Architecture Thesaurus Online, http://www.getty.edu/research/tools/vocabulary/aat/, J. Paul Getty Trust, Los Angeles, 2000
- Wikipedia: Cadmium pigments Accessed March 2026
- CHSOS: Spectra (Reflectance, XRF, Raman, FTIR) for Cadmium yellow