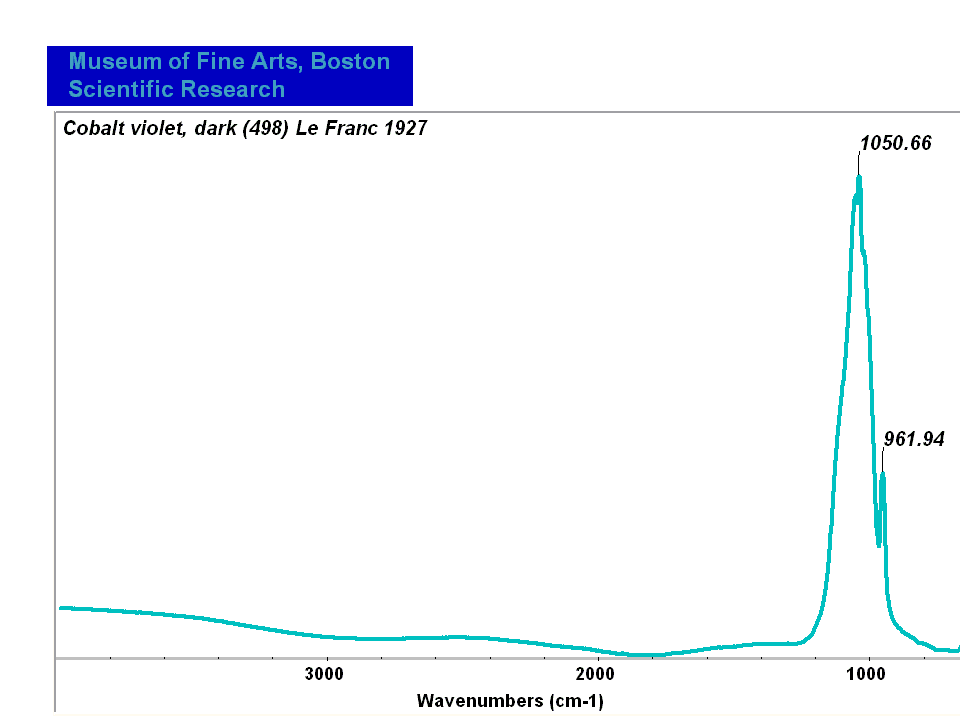
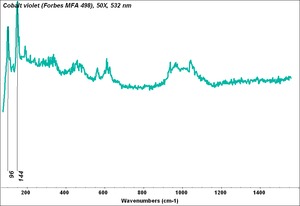
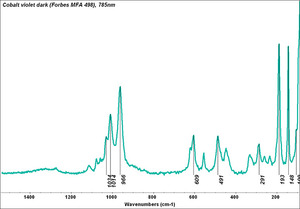
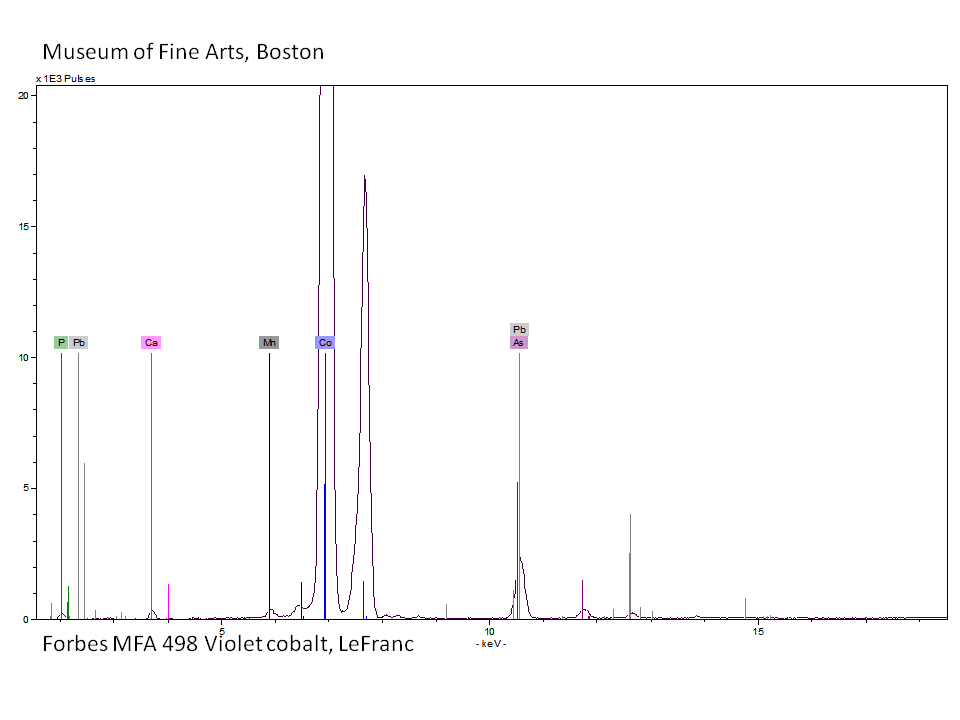
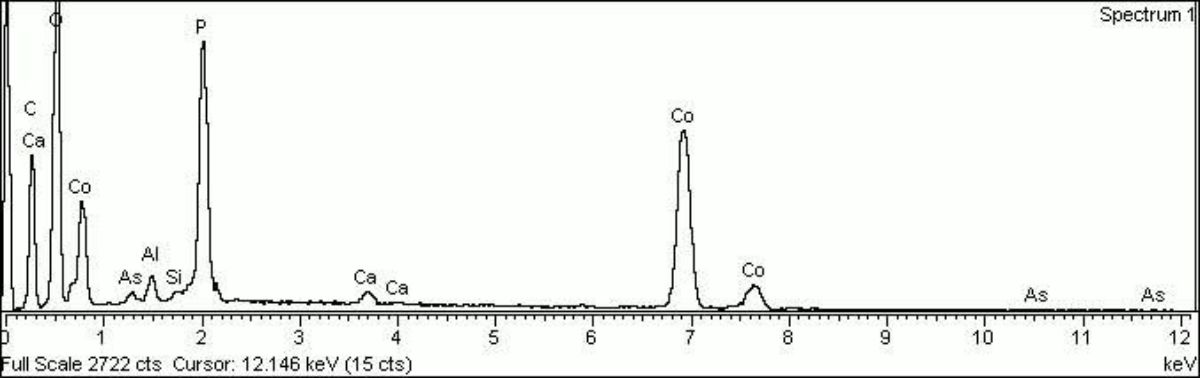
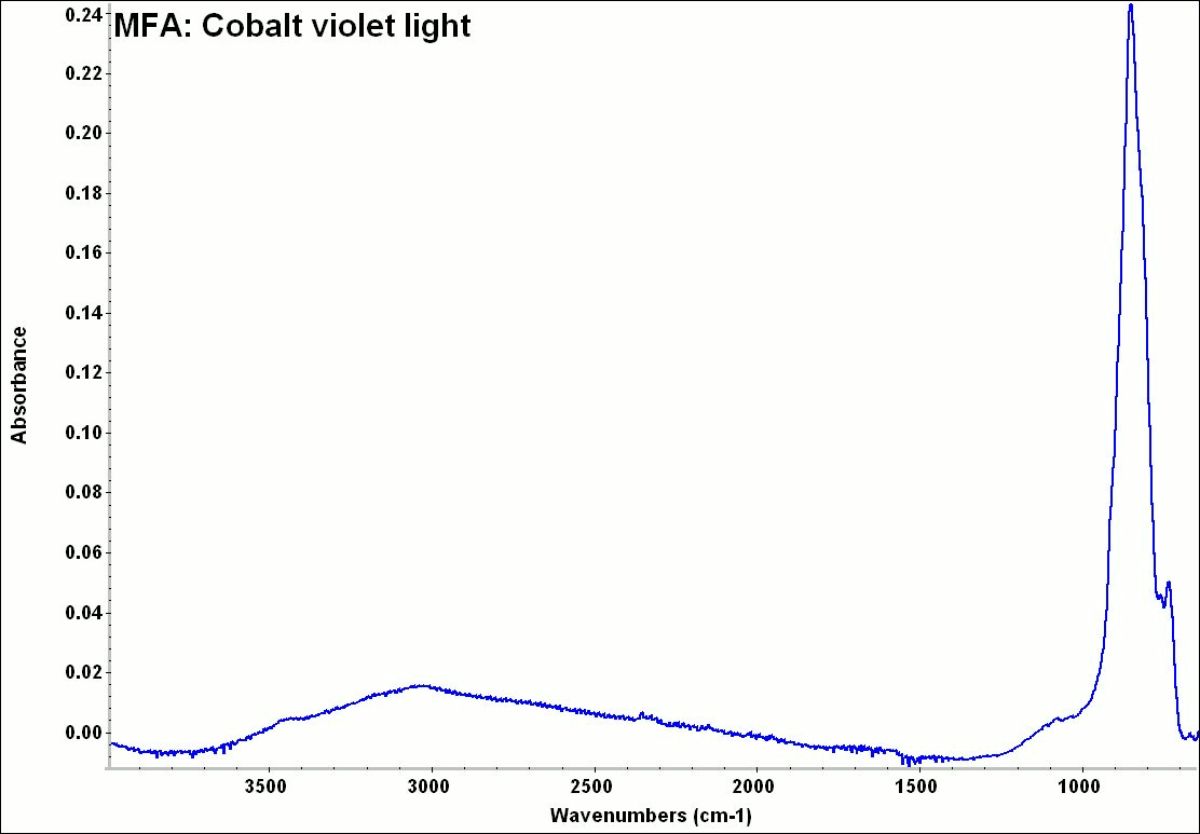
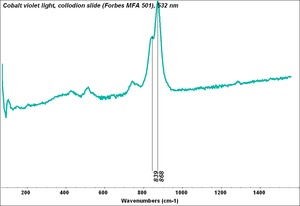
Cobalt violet
Description
A general name for several violet color cobalt pigments. First developed in 1859, cobalt violet was the primary permanent violet pigment available. Cobalt violets range from deep to pale shades with either a pink or blue hue. The first cobalt violets used were composed of cobalt arsenate. This highly toxic compound is now rarely used. Instead, most current cobalt violets are nontoxic and are made from either cobalt phosphate, or cobalt ammonium phosphate. Cobalt violets are used in paints, glass, glazes and enamels.
A pale to medium violet pigment originally composed of cobaltous arsenate. Cobaltous arsenate occurs in nature as cobalt bloom or erythrite. Once it was synthetically produced in 1880, it became an important permanent, violet pigment. Cobaltous arsenate is now rarely used because of its toxicity. It has been replaced by the use of cobaltous phosphate (deep cobalt violet) and cobaltous ammonium phosphate. Light cobalt violet was used as a colorant in paints, glass, glazes, and enamels. 2) A medium to strong violet pigment composed of cobaltous phosphate. Deep cobalt violet was first developed in 1859 by Salvetat and sold as an artists' pigment in 1890. The colorfast pigment has low tinting strength and dries quickly in oil paints.
Synonyms and Related Terms
- Cobalt violet: cobalt arsenate (light); cobalt phosphate (deep); Kobaltviolett (Deut.); violeta de cobalto (Esp.); violet de cobalt (Fr.); violetto di cobalto (It.); violeta de cobalto (Port.)
- Cobalt violet, light: Light: cobalt arsenate (arsenite); Pigment Violet 14; CI 77350; erythrite (mineral); violeta de cobalto claro (Esp.); Kobaltviolett (hell) (Deut.); violet de cobalt (clair) (Fr.); iodes toy kobaltioy anoikto (Gr.); violetto di cobalto chiaro (It.); cobalt violet (licht) (Ned.); violeta de cobalto, claro (Port.); red cobalt; cobalt bloom; ; pale cobalt violet
- Cobalt violet, deep: cobalt phosphate; deep cobalt violet; Pigment Violet 14; CI 77360; violeta de cobalto intenso (Esp.); violet de cobalt (foncé) (Fr.); Kobaltviolett (dunkel) (Deut.); iodes toy kobaltioy skoyro (Gr.); violetto di cobalto scuro (It.); cobalt violet (donker) (Ned.); violeta de cobalto, escuro (Port.)
Risks
- Skin contact may cause allergies, especially on elbows, neck and ankles.
- Chronic inhalation may cause asthma.
- Ingestion may cause vomiting, diarrhea and the sensation of hotness.
Physical and Chemical Properties
Cobalt violet, dark
- Microscopically, the particles are strongly pleochroic with colors from pink to violet to yellow.
- Highly birefringent under crossed polars.
- Appears magenta with Chelsea filter.
- Composition = Co3(PO4)2 - 8H2O
- Refractive Index = >1.662
Cobalt violet, light
- Monoclinic crystal system with prismatic or euhedral crystals.
- Perfect cleavage parallel to long axes.
- Weakly pleochroic.
- High birefringence under crossed polars.
- Composition = Co3(AsO4)2 - 8H2O
- Mohs Hardness = 1.5-2.5 (erythrite)
- Density = 3.06 g/ml
- Refractive Index = 1.626-1.701
Resources and Citations
- Pigments Through the Ages: Cobalt violet
- Corbeil, Marie-Claude, Jean-Pierre Charland, Elizabeth Moffatt. 'The characterization of cobalt violet pigments' Studies in Conservation vol.47 (2002), pp.237-249.
- R. J. Gettens, G.L. Stout, Painting Materials, A Short Encyclopaedia, Dover Publications, New York, 1966
- Ralph Mayer, A Dictionary of Art Terms and Techniques, Harper and Row Publishers, New York, 1969 (also 1945 printing)
- Encyclopedia Britannica, http://www.britannica.com Comment: "Cobalt Processing." Accessed: 15 Apr. 2004.
- Website: http://webexhibits.org/pigments/indiv/overview/coviolet.html - developed 1859
- Thomas B. Brill, Light Its Interaction with Art and Antiquities, Plenum Press, New York City, 1980
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Random House, Webster's Encyclopedic Unabridged Dictionary of the English Language, Grammercy Book, New York, 1997
- Art and Architecture Thesaurus Online, http://www.getty.edu/research/tools/vocabulary/aat/, J. Paul Getty Trust, Los Angeles, 2000
- Nicholas Eastaugh, Valentine Walsh, Tracey Chaplin, Ruth Siddall, Pigment Compendium, Elsevier Butterworth-Heinemann, Oxford, 2004
- Reed Kay, The Painter's Guide To Studio Methods and Materials, Prentice-Hall, Inc., Englewood Cliffs, NJ, 1983
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- CHSOS: Spectra (Reflectance, XRF, Raman, FTIR) for Cobalt Violet