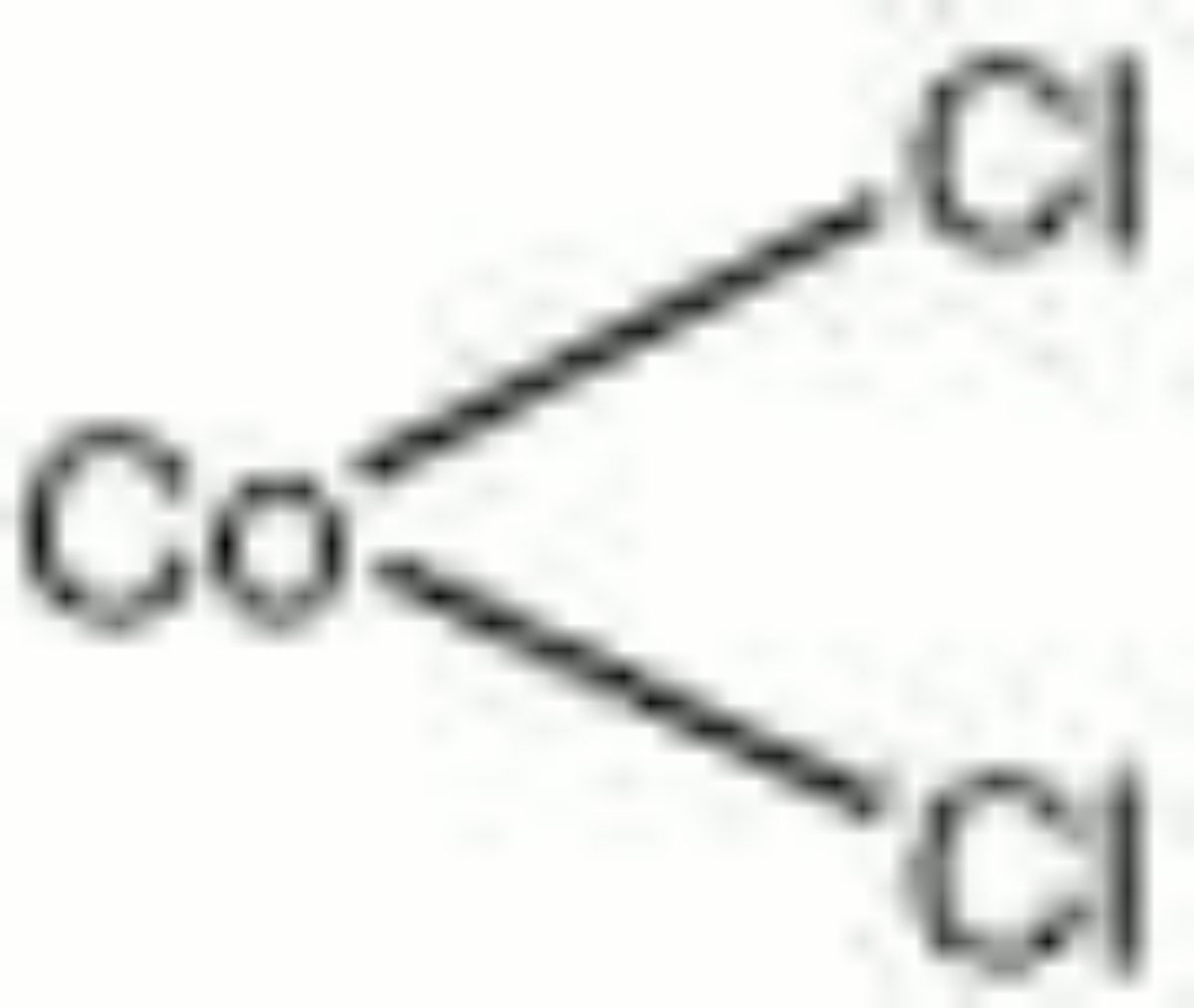
Cobaltous chloride
Jump to navigation
Jump to search
Description
Pale blue, flaky crystals. Cobaltous chloride is most commonly used as a color indicator of moisture. On exposure to moist air, anhydrous cobaltous chloride absorbs water forming pink-color hexahydrate crystals. The blue anhydrous form can be regenerated by heating at 52-56 C. Cobaltous chloride is added to silica gel and activated alumina type desiccant as a moisture indicator. It has also been used in indicator paper.
Synonyms and Related Terms
cobalt chloride; cobalt dichloride; cobalt (II) chloride; Cobalt(II)-chlorid (Deut.)
Risks
- Skin contact may cause allergies, especially on elbows, neck and ankles.
- Chronic inhalation may cause asthma.
- Ingestion may cause vomiting, diarrhea and the sensation of hotness.
- Fisher Scientific: MSDS
Physical and Chemical Properties
Soluble in water, alcohols, acetone, ether, glycerol, pyridine.
| Composition | CoCl2 |
|---|---|
| CAS | 7646-79-9 |
| Melting Point | 735 C |
| Density | 3.348 g/ml |
| Molecular Weight | mol. wt. = 129.84 |
| Boiling Point | 1049 C |
Additional Images
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 406
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Dictionary of Paper, American Paper Institute, New York, Fourth Edition, 1980
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 2498
- Wikipedia: http://en.wikipedia.org/wiki/Cobalt_chloride (Accessed Jan. 15, 2006)
- B. Gascoigne, How to Identify Prints, Thames & Hudson, London, 2004
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: ref. index=