Dayflower blue
Jump to navigation
Jump to search
Description
A blue dye extracted from the petals of the Commelina communis lily plant native to Asia. Dayflower is a herbaceous annual plant that sprawls along the ground like a vine, often rooting at the nodes. The flowers bloom only for a day, emerging one at a time. The plant is considered invasive in the United States and it is resistant to many herbicides. The blue colorant in the dayflower blue dye is primarily commelinin.
- See also [Uemera Dye Archive (Tsuki kusa)]
Synonyms and Related Terms
tsuyukusa (Jap.); aigami (Jap.); commelinin; Commelina communis (Asiatic dayflower); Commelina virginica (Virginia dayflower); mouse ears
Physical and Chemical Properties
- Leaves = alternate, narrowly ovate to lance-shaped
- Stamens = 3 on short stems (sterile) and 3 on long stems producing pollen
- Blooms = May-October
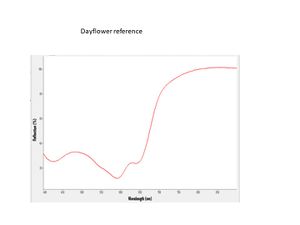
- ISO R105 Lightfastness Classification =
Resources and Citations
- S.Shimoyama, Y.Noda, S.Katshuhara, "Non-Destructive Analysis of Ukiyo-E Prints" Dyes in History and Archaeology, No.15, Paper presented in Manchester England, Nov. 1996.
- R.Feller, M.Curran, C.Bailie, 'Identification of Traditional Organic Colorants Employed in Japanese Prints and Determination of their Rates of Fading', Japanese Woodblock Prints, Allen Memorial Art Museum, Oberlin College, Oberlin, 1984
- Shiho Sasaki, Elizabeth I. Coombs, 'Dayflower blue: its appearance and lightfastness in traditional Japanese prints' in "Scientific research on the pictorial arts of Asia:proceedings of the second Forbes Symposium at the Freer Gallery of Art, 2005" AATA Getty
- Missouri Department of Conservation: Common Dayflower