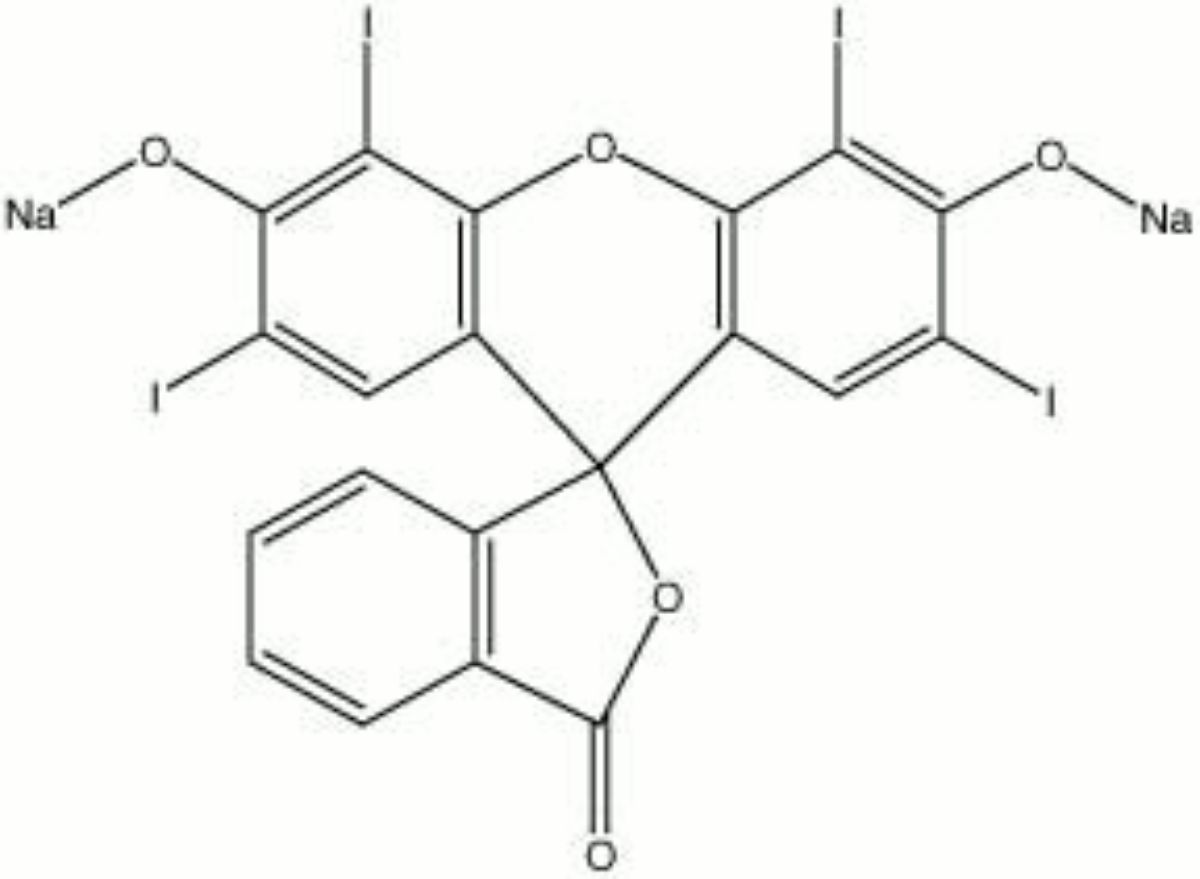
Erythrosine
Description
A brown powder that forms a cherry red synthetic dye solution with water. First discovered in 1876 by Kussamaul, erythrosine is an iodinate derivative of fluorescein. Early use of erythrosine was as a direct dye on wool and silk. It was also used in inks, lacquers, cosmetics, and as a lake pigment. However, because erythrosine was not colorfast in sunlight, its use as a textile dye was discontinued by the 1920s. It was approved as a food dye in 1906, and was popularly used in cherries, candies and cake decorating gels until it was banned in 2025 (in the U.S).
Synonyms and Related Terms
erythrosine B; erythrosine BS; Acid Red 51; CI 45430; FD&C Red No.3; Food Red 14; Pigment Red 172 (aluminum salt); Solvent Red 140; érythrosine (Fr.); eritrosina (Esp., Port.); sodium iodeosin; erythrosin
Risks
- Toxicity and carcinogenicity are being studied; lab studies show interactions with the thyroid
- Millipore Sigma: SDS
Physical and Chemical Properties
- Soluble in water, ethanol.
- An aqueous solution of erythrosine will form a yellow-brown precipitate when drops of HCl are added and a red precipitate when drops of NaOH solution are added.
- Maximum absorption wavelength = 524 nm.
- The fluorescence of erythrosine changes with pH. It is colorless at pH 4.0 changing to a fluorescent yellow-green at pH 4.5
- Composition = C20H6I4Na2O5 (mol. wt. = 879.86 g/mol.)
- CAS = 16423-68-0
Resources and Citations
- M.Ballard (ed.), Important Early Synthetic Dyes. Chemistry, Constitution, Date, Properties. Conservation Analytical Laboratory, Smithsonian Institution, 1991.
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 3734
- T.Schafer, C. Norton, V.Blyth-Hill, "The Efficacy of Using Boards Containing Zeolites in Passepartout for Works of Art on Paper, WAAC Newsletter, 22(1):14, 2000. -erythrosine fades rapidly
- Colour Index International online at www.colour-index.org Comment: discoverer = Kussmaul; CAS=16423-68-0, 12227-78-0, 15905-32-5
- CRC Handbook of Chemistry and Physics, Robert Weast (ed.), CRC Press, Boca Raton, Florida, v. 61, 1980 Comment: The fluorescence of erythrosine changes with pH. It is colorless at pH 4.0 changing to a fluorescescent yellow-green at pH 4.5
- Sigma Dyes, Stains and Natural Pigments, Infrared Library, Nicolet, 1991-1995 Comment: OMNIC for Erythrosine B: formula= C20H8I4O5, CAS= 15905-32-5 and for sodium salt of Erythrosine B: formula=C20H6I4Na2O5, CAS= 568-63-8
- Wikipedia: Erythrosine Accessed Feb. 2026