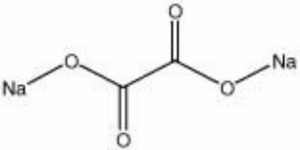
Sodium oxalate
Jump to navigation
Jump to search
Description
White, odorless hygroscopic solid. Sodium oxalate is used as a source of oxalate. It is used for finishing textiles, tanning leather, and blue printing.
Synonyms and Related Terms
ethandioic acid disodium salt
Risks
- Toxic by ingestion.
- Contact causes irritation.
- Fisher Scientific: MSDS
Physical and Chemical Properties
Soluble in water. Insoluble in ethanol.
| Composition | Na2C2O4 |
|---|---|
| CAS | 62-76-0 |
| Melting Point | 250-270 C |
| Density | 2.34 g/ml |
| Molecular Weight | mol. wt. = 134.0 |
Resources and Citations
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 736
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993
- Van Nostrand's Scientific Encyclopedia, Douglas M. Considine (ed.), Van Nostrand Reinhold, New York, 1976
- The Merck Index, Martha Windholz (ed.), Merck Research Labs, Rahway NJ, 10th edition, 1983 Comment: entry 8795