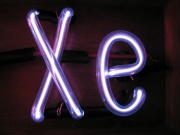Xenon
Jump to navigation
Jump to search
Description
Xenon, a noble gas element of atomic number 54, was discovered and named by William Ramsay in 1898. Under standard conditions, xenon is a heavy, colorless, odorless gas that is chemically unreactive. It is the least abundant noble gas composing 0.00087% of the atmosphere, although it occurs more frequently in gases evolved from thermal springs. The principal application for the gas is in electronic devices and lamps.
Uses: filing luminescent tubes, flash lamps for photography, lasers, anesthesia, and hermetically sealed storage cases.
Synonyms and Related Terms
Xe; xénon (Fr.); xeno (It.); Xênon (Port.); xenón (Esp.)
Risks
- Noncombustible
Physical and Chemical Properties
| Composition | Xe (atomic no. 54) |
|---|---|
| CAS | 7440-63-3 |
| Melting Point | -112 C |
| Density | 3.05 g/ml |
| Molecular Weight | atomic wt 131.293 |
| Boiling Point | -107.1 C |
Resources and Citations
- Web Elements: Website
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 652
- G.G. Hawley, The Condensed Chemical Dictionary, Reinhold Publishing, New York, 6th ed., 1961
- Encyclopedia of Chemical Technology, R.E.Kirk, D.F.Othmer (eds.), John Wiley & Sons, Inc., New York, 1984
- Wikipedia: http://en.wikipedia.org/wiki/Xenon (accessed Mar. 10, 2006)