Silica gel, commercial
Description
An amorphous material of Silica that has the ability to act as a humidity buffering agent. As a RH buffer, Silica gel absorbs moisture in humid environments and releases water in dry air. Once equilibrated to set moisture level, silica gel will maintain that humidity level in a tightly sealed environment. Silica gel performs best at room temperature (70-80F) and high humidity (60-90%). The pore size in silica gel is related to its ability to adsorb water and other pollutants. Silica gel Type A with approximately pore size of 2.5 nm is most often used for vapor-phase moisture. Silica gel saturated with water can be dried with heat (120 C for 2 hours) and used again as a desiccant. For re-use as a humidity buffer, it must be reconditioned at the required humidity.
| Product | Composition | Pre-conditioned RH options | Humidity for usage | Comments/Forms | Web page for more info |
|---|---|---|---|---|---|
| Arten Gel | type A SiO2 | 0% 30% 40% | Best for 0-40% range; avg value of MH* is 5 in range | 8 mesh beads (2mm diameter) in moisture permeable bag | APSNYC: Product page |
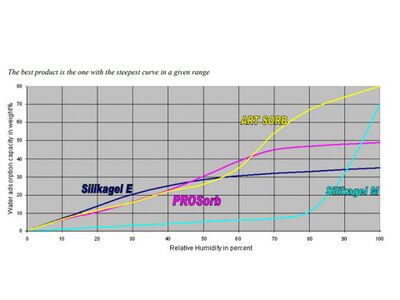
| Art Sorb | 90% SiO2 10% LiCl | 0% 40% 45% 50% 55% 60% 65% 70% | Best from 40-70% | Prosorb site says Art Sorb is best from 60-80%; beads, sheets, cassettes | Fuji Silysia: Art Sorb |
| Moisture Pak | cellulose, water, salts | 62% +/- 3% in sealed container | Same | Food storage, tobacco, wood instruments, herbs; variable size packets | Sorbent Systems: MoisturePak |
| Prosorb | 97% SiO2 3% Al2O3 | 30% 35% 40% 45% 50% 55% 60% | Optimum between 30-60% range | Similar performance as Rhapid gel; beads, sachets, cassettes | Sercalia: Prosorb |
| Rhapid gel | Hybrid SiO2 | 0% 40% 45% 50% 60% | Best for 0-65% range; avg value of MH* is 5 in range | Flexible moisture-pearmeable sachets | APSNYC: Products page |
| Silica gel E | type A SiO2 | Best for 0-30% | |||
| Silica gel M | Macroporous SiO2 | Best for 80-100% | Prevents condensation at high RH | ||
| Silica gel orange | type A SiO2 with either methyl violet or iron salts as a moisture indicator | Best for 0-30% | Moisture indicator: methyl violet changes from orange to green;iron salts change orange/brown to colorless; | AGM: Blue vs orange | |
| Grade 42 | type A SiO2 with cobalt dichloride indicator | Moisture indicator: changes blue to pink as RH increases from 15-45% | |||
| Grade 40 | type A SiO2 | may be mixed with 42 |
Moisture Reservoir Information
The moisture adsorbing properties of silica gels are affected by factors such as capillary pore size or the inclusion of hygroscopic salts, resulting in a wide range of performance. Thus, it is important to compare the buffering capacity of different types of silica gels to determine which has the best performance for a specific application. In 1977, Thomson described the specific moisture reservoir with the variable M where M represents the amount of water (in grams) that is gained or lost by 1 kilogram of silica gel for each 1% change in RH. However for any specific silica gel type, M can vary widely due to the expected RH range and the hysteresis of the adsorption/desorption curve. In order to compensate, a new value called MH has been defined as the average amount of water (in grams) that is gained or lost by 1 kilogram of silica gel for each 1% change in RH. This is determined by repeatedly cycling silica gel between adsorption and desorption within a specific RH range until a constant value is measured.
TABLE: Specific moisture reservoir (g/kg for a 1% RH change) at 20°C *
| Moisture sorbent | MH 50 ± 10% | M 20 → 30% | M 60 → 50% | Recommended amounts |
|---|---|---|---|---|
| RH around 50% | Keep RH low | Keep RH high | Depends on air flow and exhibit length | |
| RHapid Gel | 6.11 ± 2.16 | 4.48 ± 0.42 | 4.38 ± 1.40 | 2-4 kg/m3 |
| PROSorb | 5.42 ± 1.32 | 4.37 ± 0.10 | 4.25 ± 0.38 | 2-4 kg/m3 |
| Art-Sorb | 4.04 ± 0.80 | 2.84 ± 0.47 | 4.18 ± 0.49 | 4-8 kg/m3 |
| Arten Gel | 5.7 | 5.5 | 9-18 kg/m3 | |
| Regular density silica gel (clear, type A) | 1.93 ± 0.44 | 5.48 ± 0.40 | 1.47 ± 0.16 | 9-18 kg/m3 |
| Orange silica gel | 1.16 ± 0.26 | 4.92 ± 0.59 | 0.94 ± 0.14 | 9-18 kg/m3 |
| Bentonite clay (Desi Pak) | 1.19 ± 0.07 | 2.25 ± 0.03 | 1.11 ± 0.02 | |
| Molecular sieves 4A (zeolite) | 0.33 ± 0.02 | 0.47 ± 0.04 | 0.31 ± 0.01 |
Table from: J. Tetreault (2017) with additional information from S. Weintraub (2002)
Resources and Citations
- AIC Conservation Wiki: Exhibit Technical Notes: Environmental Control
- Exhibit Guidelines Technical Note: Silica Gel Products and How to Use Them
- Exhibit Guidelines Technical Note: Conditioning Silica Gel
- Exhibit Guidelines Technical Note: Conditioning Silica Gel with a Saturated Salt Solution
- J.Tétreault, P.Bégin, Silica Gel: Passive Control of Relative Humidity, CCI Technical Bulletin no. 33. Canadian Conservation Institute. Ottawa (2017).
- Art Preservation Services: Silica Gel Technical Information
- S. Weintraub, "Demystifying silica gel", Objects Specialty Group Postprints, Vol.9, p. 169-194, 2002 Link
- G. Thomson, "Stabilization of RH in Exhibition Cases: Hygrometric Half-time", Studies in Conservation 22: 85-102, 1977.