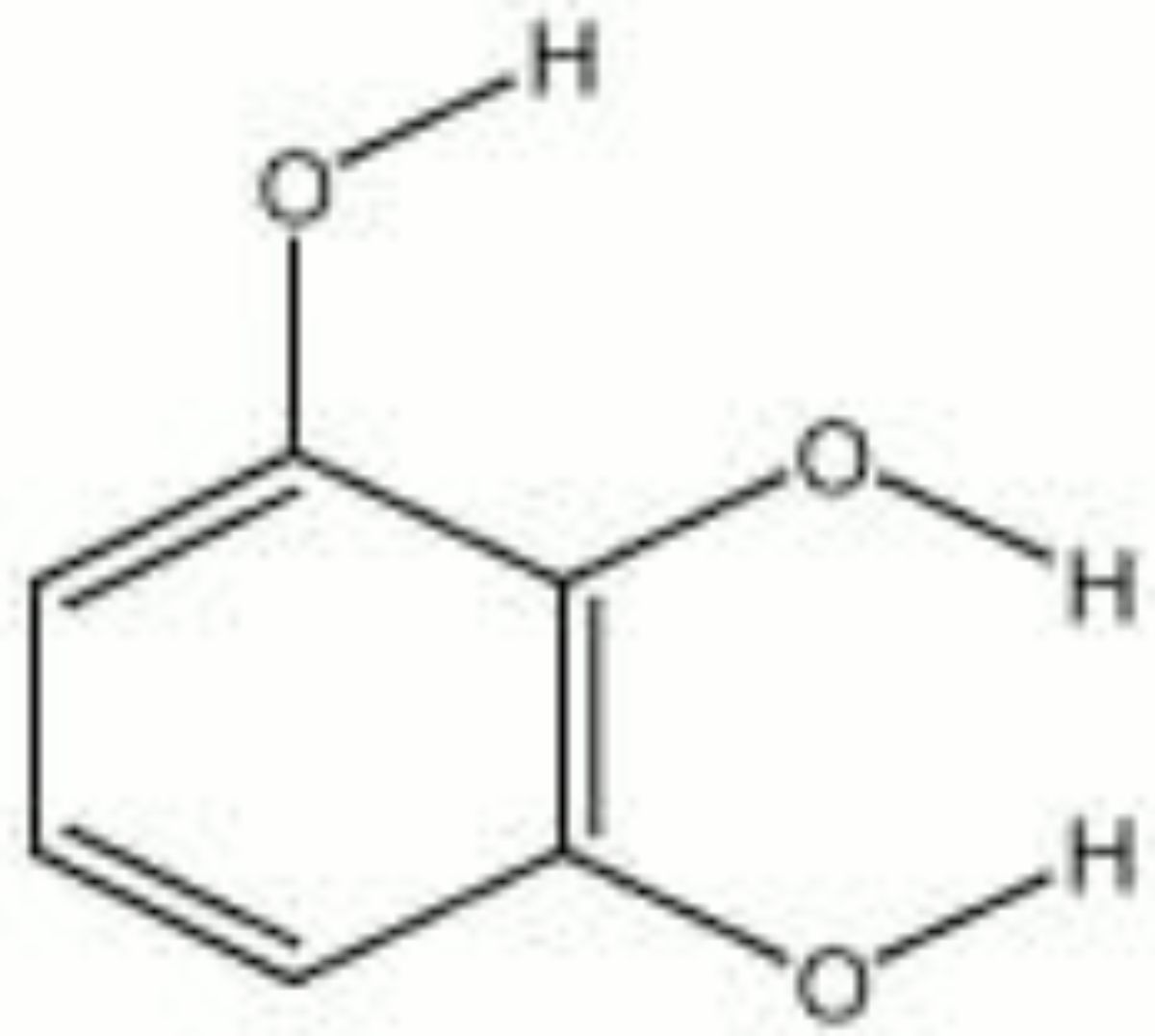
Pyrogallic acid
Jump to navigation
Jump to search
Description
Tiny, white, light-sensitive crystals that tend to float with air movement. Pyrogallic acid, or pyro, is used as a developer in photographic solutions. It reduces the salts of gold, silver, mercury, and platinum to their metallic state. This characteristic was first noted in 1832 and soon after it was applied to photograph development. Pyrogallic acid is also used in the manufacture of dyes.
Synonyms and Related Terms
pyrogallol; pyro; 1,2,3-trihydroxybenzene; 1,2,3-benzenetriol; Fouramine brown AP; Fourrin; CI 76515; Oxidation base 32; Piral
Risks
- Toxic by ingestion, inhalation and skin absorption.
- Skin contact can cause irritation and allergies.
- Combustible.
- Fisher Scientific: SDS
Physical and Chemical Properties
Soluble in water, ethanol, ether. Needle-shaped crystals.
| Composition | C6H3(OH)3 |
|---|---|
| CAS | 87-66-1 |
| Melting Point | 131-134 C |
| Density | 1.45g/ml |
| Molecular Weight | mol. wt.=126.1 |
| Boiling Point | 309 C |
Resources and Citations
- Matt Roberts, Don Etherington, Bookbinding and the Conservation of Books: a Dictionary of Descriptive Terminology, U.S. Government Printing Office, Washington DC, 1982
- Michael McCann, Artist Beware, Watson-Guptill Publications, New York City, 1979
- G.S.Brady, Materials Handbook, McGraw-Hill Book Co., New York, 1971 Comment: p. 796
- Richard S. Lewis, Hawley's Condensed Chemical Dictionary, Van Nostrand Reinhold, New York, 10th ed., 1993